Why valency of calcium is +2?
In calcium oxide, the calcium is electropositive in nature and to fulfil its octet, the calcium atoms lose two electrons to form the calcium cations whose valency is 2, hence the corresponding anion, i.e. the oxide anion must also have valency 2 to balance the positive charge on the calcium cation.
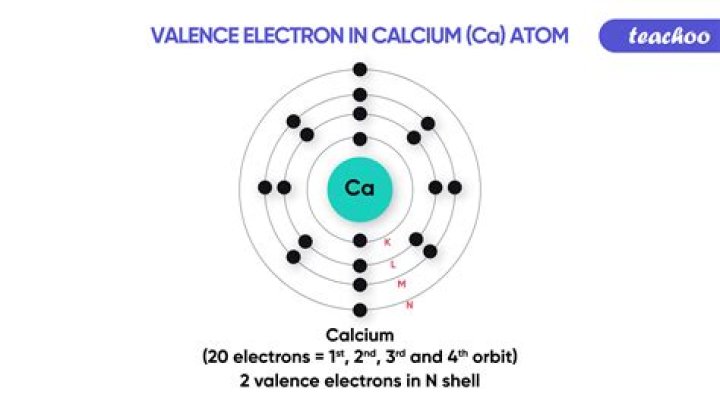
How do you find the valency of calcium?
Thus the valency of calcium is 2 since it has 2 valence electrons. 2. If in the valence shell, the number of electrons is > 3, the atom is of a non metal. To find the valency of a non metal, subtract the number of valence electrons from 8.
What is valency of calcium CaCO3?
The valency of calcium is +2 in the compound CaCO3 or Calcium Carbonate. Calcium atom has two free electrons in its outermost shell that is easily transferred to the CO3 or carbonate group.
What are the valencies of Ca and CL?
Hint:The valency of Calcium is 2 and chlorine is 1. And thus 2 atoms of chlorine will be required to bond with 1 atom of calcium so that octet of both the species is completed and a stable compound is formed. All the atoms try to get octet in order to gain extra stability.
What is the valence of antimony?
Antimony contains 5 electrons in its outer shell, thus it has 5 valence electrons. The electronic configuration of Antimony is 2, 8, 18, 18, 5.
What is chlorine valency?
This shows chlorine has 7 valence electrons in its outermost shell. If the valence electron is more than 4, then the valency can be calculated as follows: Valency = Valence electron -8. Accordingly, the valency of chlorine is 7-8 which is 1.
What is the valency of calcium Ca in CaCl2?
Hi, Valency of calcium is +2, thus to balance the charge, two chlorine atoms combine each having -1 charge, a total of -2 charge and hence neutralising the whole charge.
What is the valency of calcium in CaO?
Ca has a valency of 2 in the combination CaO.
What is the valency of CL in CaCl2?
Explanation: CaCl2:- Valency of Calcium is 2 and Valency of Chlorine is 1.
What is the valence of Krypton?
Explanation: Since Kr or Krypton is a noble gas, it has a full valence shell or octet of 8 electrons.
How many valence electron Does CA have?
A: Calcium is a group 2 element with two valence electrons.



