Why is phosphorus written P4?
Answer: Phosphorus can form a P4 white phosphorus tetrahedron because it can form three bonds. It can make a tetra-atomic P4 molecule by sharing valency electrons with three other P atoms to complete its octet. …
Why does phosphorus exist as P4 but nitrogen as n2 gas?
Nitrogen thus forms a very stable diatomic molecule, N2. On moving down a group, the tendency to form pπ-pπ bonds decreases (because of the large size of heavier elements). Therefore, phosphorus (like other heavier metals) exists in the P4 state.
What does P4 mean in chemistry?
Phosphorus molecule (P4)
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Earth.
Why is P4 a molecule?
The phosphorus is so larger that the p-orbitals which are required to overlapp sideways , are unable to overlapp sufficiently to make a pie bond. But phosphorus have a tendency to show catenation. And because of catination it can join with its own atoms and make covalent bonds. and thus it makes P4 molecule.
N2 is exist as diatomic molecule in which two nitrogen atoms are attached to each other by triple bond this triple bond is due to p(pie)-p(pie) multiple bonding (sidewise overlapping) and such bonding is not possible in phosphorus due to it’s large size but possible in nitrogen due to it’s small size.
Why does N2 show Catenation property less than P4?
Since nitrogen atom is smaller, there is greater repulsion of electron density of two nitrogen atoms, thereby weakening the N-N single bond.
Is P4 a molecule or atom?
P4 is a substance that is made up of four atoms of the same element, so it is a molecular element.
Nitrogen exists as diatomic molecule and phosphorus as P4 .
Is P4 a gas?
N2 is gas But p4 is solid – YouTube.
Why is phosphorus named phosphorus?
The name derives from the Greek phosphoros for “bringing light” because it has the property of glowing in the dark. This was also the ancient name for the planet Venus, when it appears before sunrise. Phosphorus was discovered by the German merchant Hennig Brand in 1669.
What is the purpose of phosphorus?
What is phosphorus? Phosphorus is a mineral found in your bones. Along with calcium, phosphorus is needed to build strong healthy bones, as well as, keeping other parts of your body healthy.
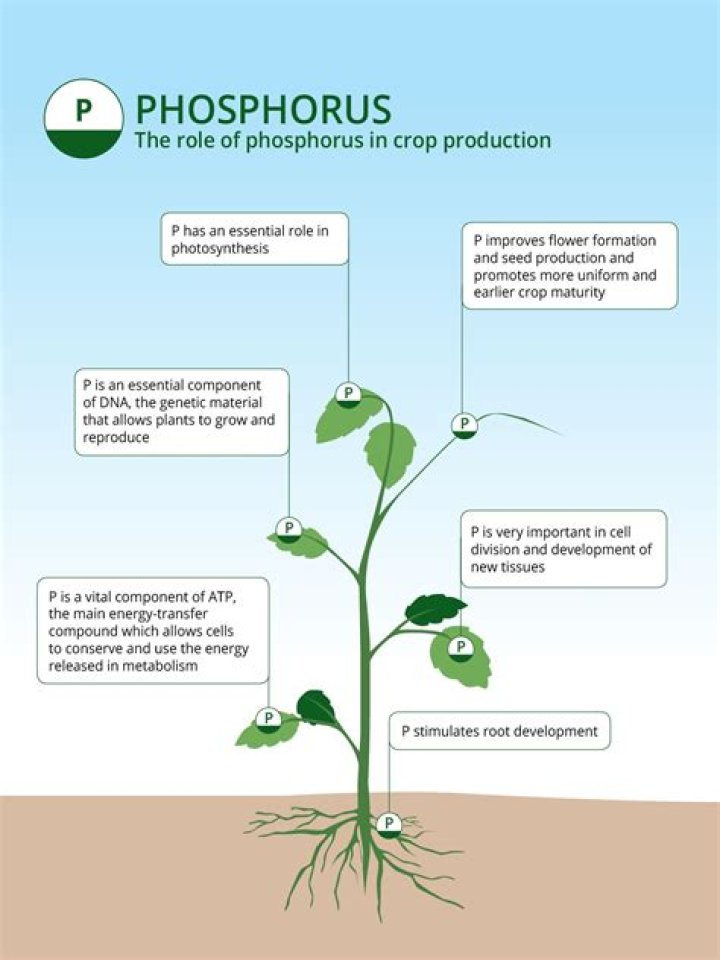
Phosphorus is essential to all living things. It forms the sugar-phosphate backbone of DNA and RNA. It is important for energy transfer in cells as part of ATP (adenosine triphosphate), and is found in many other biologically important molecules.
What bond is P4?
The Phosphorus P4 molecule contains a total of 3 bond(s) There are 3 non-H bond(s), 3 multiple bond(s), 1 double bond(s) and 2 triple bond(s). The 2D chemical structure image of Phosphorus P4 is also called skeletal formula, which is the standard notation for organic molecules.
Why does P4 exist and not n4?
Due to larger atomic size P is unable to form pi bonds and so it is tetra-atomic in which each P atom is linked with 3 other P atoms by 3 sigma bond. But, due to smaller atomic size N forms 1 sigma and 2 pi bonds i.e. triple bonds with other N atom and exists as diatomic molecule.
Why is P4 tetrahedral?
The shape of P4 is tetrahedral. Each P atom makes 3 bonds and has a lone pair, making each formal charge 0.
Molecular nitrogen is a colorless, odorless, tasteless, and inert gas at normal temperatures and pressures. About 78% of Earth’s atmosphere is nitrogen. The strong triple-bond between the atoms in molecular nitrogen makes this compound difficult to break apart, and thus nearly inert.
Why should it make sense that N2 exists as a gas?
Hydrogen bonding is the bonding is a special dipole-dipole interaction between a hydrogen atom in a polar bond with either N,O, or F atom. Why should it make sense that N₂ exists as a gas? Lowering the temperature would cause the nitrogen to undergo a phase change from gas to liquid.
Why is nitrogen found in a gaseous state?
Nitrogen is found in a diatomic form with a triple bond between two atoms. N2 molecules are held together by weak van der Waals force of attraction which can be easily broken by the collision of the molecules. So N2 is a gas at room temperature.



