Why is nitrogen triiodide polar?
Nitrogen triiodide is most polar because the molecule is asymmetric, while. Oxygen is nonpolar because both atoms have identical electronegativities. Nitrogen triiodide is most polar because the molecule is asymmetric, while carbon disulfide is. Oxygen is nonpolar because both atoms have identical electronegativities.
Is nitrogen triiodide polar or non polar?
Nitrogen triiodide is most polar because the molecule is asymmetric, while carbon disulfide is symmetric, canceling out the dipoles of the bonds. Silicon tetrafluoride has a structure identical to methane, except that carbon is replaced with silicon and hydrogen replaced with fluorine.
Does nitrogen cause polarity?
Bonds between carbon and other elements such as oxygen and nitrogen are polar. The polarity of a bond depends on the electronegativities of the bonded atoms. Large differences between the electronegativities of the bonded atoms increase the polarity of bonds.
And that the Lewis structure of Silicon dioxide does not have any lone pairs of electrons. So there is no shape distortion, making it easy to cancel out the dipole moment produced by the Si-O bond. As a result of all of these factors, SiO2 is a non-polar molecule.
How are polar molecules different from nonpolar?
Polar molecules occur when there is an electronegativity difference between the bonded atoms. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out.
The hybridization of NI3 is Sp³. The molecular geometry of NI3 is trigonal pyramidal and its electron geometry is tetrahedral. Lewis structure of NI3 contains 1 lone pair and 3 bonded pairs.
Why is nitrogen triiodide so unstable?
The reason nitrogen triiodide is so unstable is down to its molecular structure – one nitrogen atom and three iodine molecules all on one side due to how the nitrogen’s electrons are arranged. And all that repelling causes something called bond strain, which makes the entire molecule extremely unstable.
What type of reaction is nitrogen triiodide?
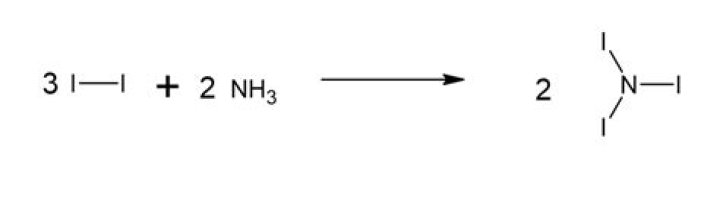
The process 2 NI3 (s) N2 (g) + 3 I2 (s) is exothermic, so that N2 (g) + 3 I2 (s) 2 NI3 (s) is endothermic. Endothermic compounds tend to be unstable.
Is SiBr4 polar or non-polar? ۔ SiBr4 is non-polar the charge is canceled in the center of the balanced molecule.
Is CS2 polar or nonpolar?
CS2 C S 2 is a nonpolar molecule is a nonpolar molecule . Explanation: CS2 C S 2 , similarly to CO2 C O 2 , is a linear molecule
Is NI3 ionic or covalent?
The chemical formula NI3 is named nitrogen triiodide. It’s a covalent molecule, which means the atoms in the molecule share electrons.
Is sbr2 polar or nonpolar?
Geometry: Bent
However, their values are not totally equal so there will still be a net dipole moment. But this dipole moment is relatively small compared to the net dipole moment of molecules that have the same geometry. SBr2 is only slightly polar, its polarity is so small that is considered non-polar.
It has zero dipole moment as the two nitrogen atoms present in nitrogen molecules have the same electronegativity hence zero electronegativity difference. So, it is true that the nitrogen molecule is a non-polar and a covalent molecule.
Is nitrogen reactive or nonreactive?
Nitrogen is a rather unreactive element, and the reason is that the N≡N bond energy is 946 kJ mol−1. This lack of reactivity is somewhat unlike other nonmetals given the position of the atom in the periodic table and the fact that nitrogen is a nonmetal having an electronegativity of 3.0 (the third highest value).
What makes a molecule polar?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A dipole is any molecule with a positive end and a negative end, resulting from unequal distribution of electron density throughout the molecule.



