Why is ncl3 polar and BCl3 nonpolar?
Boron Trichloride or BCl3 is a nonpolar compound because of its symmetrical structure ie; Trigonal Planar. The B-Cl bond itself is polar because of the difference in electronegativity of Boron(2.04) and Chlorine(3.16) atoms and all three B-Cl bonds lie at 120 degrees to each other.
Is BCl3 or NCl3 more polar?
The BCl3 is a planar molecule whereas NCl3 is pyramidal becausea B-Cl bond is more polar than N-Cl bondb N-Cl bond is more covalent than B-Cl bondc nitrogen atom is smaller than boron atomsd BCl3 has no lone pair but NCl3 has a lone pair of electrons.
What is NCl3 polar or nonpolar?
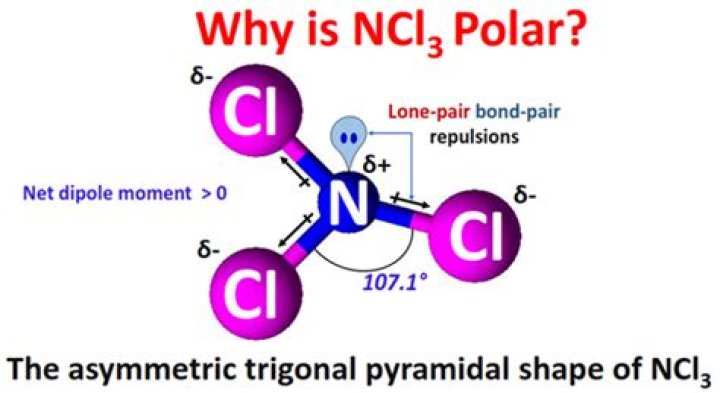
NCl3 N C l 3 is a polar molecule.
All trigonal pyramidal molecules are polar because the asymmetry introduced by the lone pair ensures the bond dipoles do not cancel out to zero. When bond dipoles do not cancel out, there is a resulting net dipole that makes the molecule polar.
Polar molecules occur when there is an electronegativity difference between the bonded atoms. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out.
Is BCl3 ionic or covalent?
Boron and chlorine are both nonmetals, so this is a covalent compound.
a. The compound NCl3 is an ionic compound (metal and nonmetal), and therefore does not require prefixes- -so NCl3 is nitrogen trichloride. b. The compound AlCl3 is an ionic compound (metal and nonmetal), and therefore does not require prefixes- -so AlCl3 is aluminum chloride.
Is N2 polar or nonpolar?
So, is N2 polar or nonpolar? N2 is a nonpolar molecule because of its linear geometrical structure and it is a diatomic molecule. As a result, both atoms have equal electronegativity and share an equal proportion of charge and the overall molecule result in a net-zero dipole moment making it a nonpolar molecule.
What type of intermolecular force is NCl3?
I will be grateful for the explanation on why NCl3 has a dipole-dipole intermolecular force, if, based on electronegativity difference, or rather the absence of such, (both N and Cl have 3.0 electronegativity) this is a non-polar bond?
Nitrogen triiodide is slightly polar in nature. The molecular geometry of NI3 is trigonal pyramidal and its electron geometry is tetrahedral.
Which of the following has maximum dipole moment a NCl3 B NBr3 C NH3 D NI3?
Out of NBr3, NI3 and NCl3, the dipole moment will be maximum for NI3, as I is most electronegative.
Is ammonia polar or nonpolar?
Ammonia is polar, the N is the negative end, and the middle of the H’s is the positive end.



