Why is H2CO sp2 hybridized?
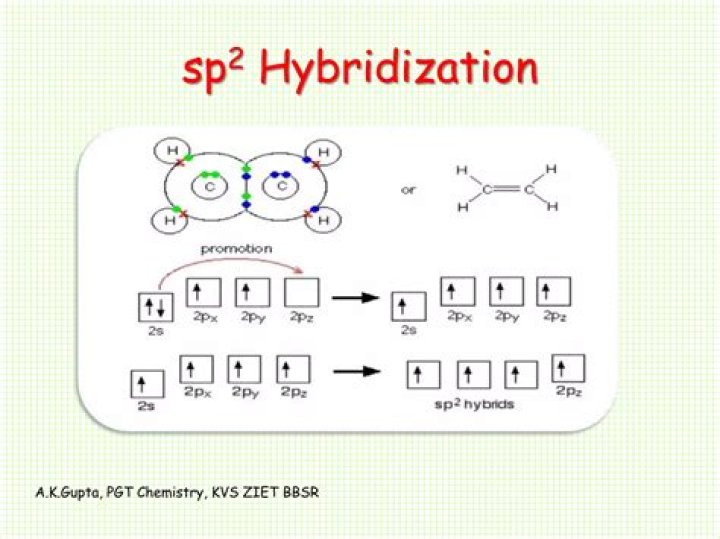
H2CO; C is sp2 hybridized.
Why is H2CO sp2 hybridized?
For example, given H2CO, the C has an sp2 hybridization because it only goes it 3 directions; two to H’s and a double bond to O. Visualize this by drawing the Lewis Structure of H2CO and looking at the directions.
Is CH2O sp2 hybridized?
CH2O is sp2 hybridized. The double bond consists of one sp hybrid orbital and one p orbital on both atoms. The lone pairs occupy the other two sp orbitals on Oxygen.
What is H2CO molecular geometry?
The central atom of H2CO, has three regions of electron density around it. The regions of electrons making a trigonal planar shape, giving a bond angle of 120掳. All three of these regions of electrons are bonding so the arrangement of the bonds/molecular shape is trigonal planar.
Why is h2o sp3 hybridized?
The central atom here is oxygen which is hybridized. So if we observe the formation of the water molecule there are three 2p orbitals and one 2s orbital. These combine to create the four sp3 hybrid orbitals.
How many electron pairs are in H2CO?
There are a total of 4 lone pairs of electrons and 8 shared pairs of electrons present in the lewis dot structure of H2CO.
What shape is CH2O?
The VSEPR model predicts that CH鈧侽 is trigonal planar with bond angles of about 120 掳.
How many electron group is CH2O?
The molecular geometry of CH2O is trigonal planar. The electron geometry of CH2O is also trigonal planar. There are a total of 4 lone pairs of electrons and 8 shared pairs of electrons present in the lewis structure of CH2O. The net dipole moment of formaldehyde is 2.330 D.
Is H2CO asymmetric or symmetrical?
Thus H2CO is an almost symmetric top and its rotational spectrum should resemble that of a symmetric top. However, each symmetric top transition will be asymmetry-split into several resolvable transitions.
Why is H2CO polar?
H2 CO is polar because the shared electrons between the carbon atom and the oxygen atom are not shared equally between the two.
What is the hybridization of NH2?
The hybridization and shape of NH2 ion are a sp2 and class 11 chemistry JEE_Main.
What is the hybridization of H?
Hydrogen never hybridizes. It’s impossible because it only has one electron, so it can never make more than one bond in normal molecules.
What is hybridization of so2?
In sulphur dioxide, the hybridization that takes place is sp2 type. To determine this, we will first look at the sulphur atom which will be the central atom. During the formation of SO2, this central atom is bonded with two oxygen atoms and their structure can be represented as O=S=O.
What is the dipole moment of H2CO?
The two C-O bond moment direction is opposite to each other and cancel each other as a result net dipole moment is zero鈥 Basiclly CH2O is known as Formldehyde (Common name) or Methanal (IUPAC). Basiclly CH2O is known as Formldehyde (Common name) or Methanal (IUPAC).
Which atom is the central atom for H2CO?
The central atom here is carbon that has three electron density regions around it. This configuration makes a trigonal planar shape with bond angles



