Why Calcium chloride is ionic?
By Liam Parker
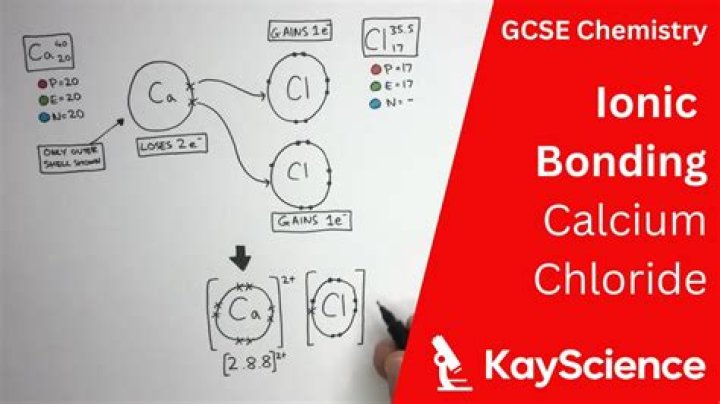
CaCl2 is an ionic compound owing to the large electronegativity difference between the calcium atom and chlorine atom, which is greater than 2.0. In calcium chloride, the calcium atom donates its two electrons and become cation whereas each chlorine atom gain one electron, donated by Calcium, and get a negative charge.



