Which type of molecule is sugar polar or nonpolar?
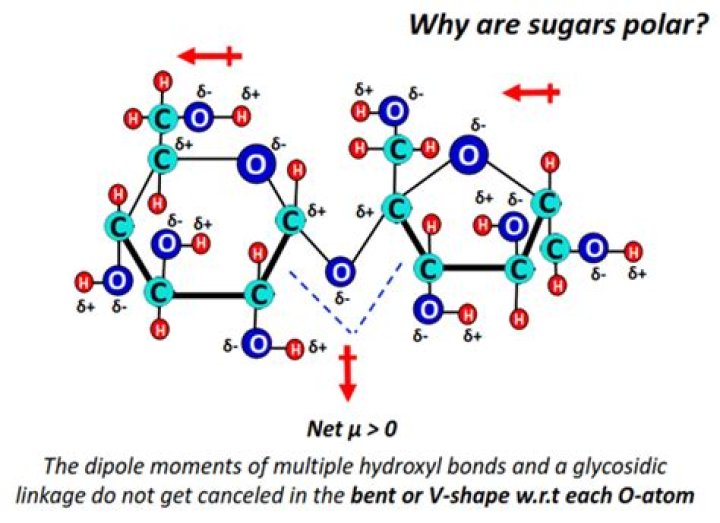
Sugar is a polar substance. This is because it contains several polar OH groups which are comprised of a highly electronegative oxygen atom bonded…
Is a sugar molecule polar or nonpolar?
The bond between the oxygen and hydrogen atoms (O–H bond) in sugar (sucrose) gives the oxygen a slight negative charge and the hydrogen a slight positive charge. Sucrose is a polar molecule.
Is sugar and salt polar?
It’s almost the same. In my opinion, it is different because sugar and water are polar substances, so sugar can dissolve easily. However, salt can be polar and nonpolar, so it may be difficult to dissolve in water. Polar substances dissolve in polar substances.
Individual sugar molecules, the monosaccharides, can be used as monomers joined together to form larger structures. For example, two glucose molecules can be joined to form the disaccharide called maltose,. Or two different sugars (fructose and glucose) can be joined together to form the disaccharide sucrose.
What are sugar atoms?
Sugar molecules are composed of carbon, oxygen, and hydrogen atoms.
Sugar, sucrose, or table sugar is a compound. The reason behind this fact is that sugar is composed of carbon, hydrogen, and oxygen atoms and a single sugar molecule contain 12 atoms of carbon, 22 atoms of hydrogen, and 11 atoms of oxygen.
Is sugar a compound or homogeneous mixture?
Sugar is a compound that is formed by a combination of three atoms: carbon, hydrogen and oxygen. Since these three atoms are chemically joined with each other thus they form a compound in nature.
Is glucose a sugar molecule?
Glucose is a simple sugar or monosaccharide. It’s your body’s preferred carb-based energy source ( 1 ). Monosaccharides are made up of one single unit of sugar and thus cannot be broken down into simpler compounds. They’re the building blocks of carbohydrates.
Chemically, sugar consists of carbon (C), oxygen (O), and hydrogen (H) atoms, and is classified as a carbohydrate . There are three main groups of sugars, classified according to the way the atoms are arranged together in the molecular structure.
Is sugar a conductor?
No, sugar solution does not conduct electricity. Sugar solution does not contain free ions that are required to conduct electricity. Sugar molecules are held by covalent bonds, as a result, they do not dissociate free ions in water.
Does sugar dissociate in water?
* Note: sugar does not dissociate in water; it does not form ions. Solid Silver (I) Chloride. What is Dissolving ? When a covalent compound (eg sugar) dissolves in water, the molecules mostly disperse; they do not all dissociate.
Is sugar soluble in ethanol?
Sugar does not dissolve very well in alcohol because alcohol has a large part that is pretty non-polar.
Sugar dissolves faster in hot water than it does in cold water because hot water has more energy than cold water. When water is heated, the molecules gain energy and, thus, move faster. As they move faster, they come into contact with the sugar more often, causing it to dissolve faster.
Does sugar have a high solubility?
You should have noticed sugar had the highest solubility of all your tested compounds (about 200 grams per 100 milliliters of water) followed by Epsom salts (about 115 grams/100 milliliters) table salt (about 35 grams/100 milliliters) and baking soda (almost 10 grams/100 milliliters).
Is sugar dissolving in water physical or chemical?
Sugar dissolving in water is a physical change. It is because no new substance is formed. Also, the process is reversible – water and sugar can be separated by vaporization followed by condensation and crystallization.



