Which term describes atoms with different masses due to varying numbers of neutrons?
Often, elements come in both radioactive and nonradioactive versions that differ in the number of neutrons they contain. These different versions of elements are called isotopes, and small quantities of radioactive isotopes often occur in nature.
Which term describes atoms with different masses due to varying numbers of neutrons?
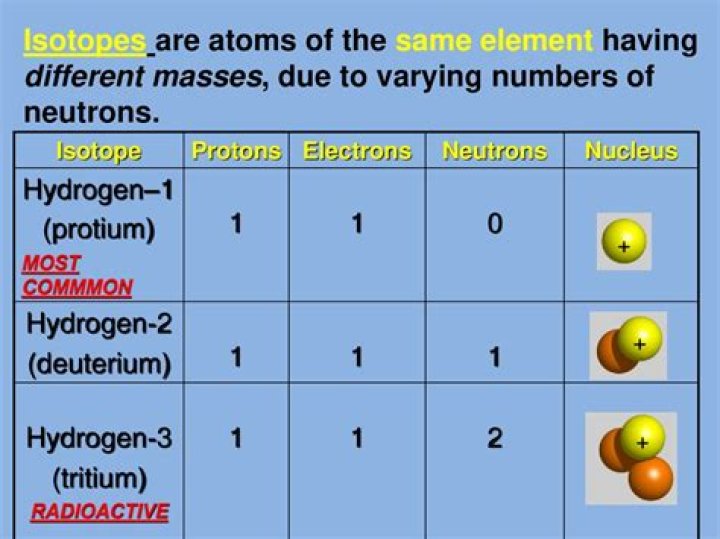
Two atoms with the same atomic number, but different mass numbers (same number of protons, different number of neutrons), are called isotopes, or isotopic nuclides. Having different numbers of neutrons changes the mass of these atoms, so isotopes have slight variations in their physical and chemical behavior.
Which term is defined as the mass of an atom based on the mass of an atom of carbon-12?
atomic mass unit: Abbreviated as 鈥渁mu.鈥 A mass equal to one twelfth the mass of an atom of carbon-12.
What are elements with different atomic masses called?
Isotopes are atoms with different atomic masses which have the same atomic number. The atoms of different isotopes are atoms of the same chemical element; they differ in the number of neutrons in the nucleus.
What is the atomic mass of a carbon isotope that has 6 protons and 7 neutrons?
Carbon atoms with 7 neutrons have an atomic mass of 13 (6 protons + 7 neutrons = 13). These atoms are the isotope called carbon-13.
What is also known as an element that has the same number of protons but different number of neutrons?
Isotopes. An isotope is one of two or more forms of the same chemical element. Different isotopes of an element have the same number of protons in the nucleus, giving them the same atomic number, but a different number of neutrons giving each elemental isotope a different atomic weight.
What has the same atomic number but different atomic masses?
Isotopes: The elements which have the same atomic number but different atomic mass, are known as isotopes. For example: carbon鈭12 and carbon鈭14.
How is atomic mass defined?
atomic mass, the quantity of matter contained in an atom of an element. It is expressed as a multiple of one-twelfth the mass of the carbon-12 atom, 1.992646547 脳 10鈭23 gram, which is assigned an atomic mass of 12 units. In this scale, 1 atomic mass unit (amu) corresponds to 1.660539040 脳 10鈭24 gram.
Why is atomic mass of an atom defined as relative atomic mass?
The atomic mass constant (symbol: mu) is defined as being 112 of the mass of a carbon-12 atom. Since both quantities in the ratio are masses, the resulting value is dimensionless; hence the value is said to be relative.
What is the atomic mass of the element?
The atomic mass of an element is the average mass of the atoms of an element measured in atomic mass unit (amu, also known as daltons, D). The atomic mass is a weighted average of all of the isotopes of that element, in which the mass of each isotope is multiplied by the abundance of that particular isotope.
How is mass number different from atomic number?
Atomic mass is associated with the number of neutrons and protons that are present in a particular nucleus of an element. Atomic number is usually the number of protons present in an element’s nucleus. It is the average weight of an element.
Which of these refers to atoms with the same atomic number but different atomic masses quizlet?
Atoms with the same atomic number and different atomic masses are referred to as isotopes.
Are atomic mass and atomic weight the same?
Atomic mass (ma) is the mass of an atom. A single atom has a set number of protons and neutrons, so the mass is unequivocal (won’t change) and is the sum of the number of protons and neutrons in the atom. Atomic weight is a weighted average of the mass of all the atoms of an element, based on the abundance of isotopes.
What has 6 protons and 6 neutrons and 6 electrons?
That means a carbon atom has 6 protons, 6 neutrons, and 6 electrons.
What is the atomic mass of an atom that has 6 protons 6 neutrons and 6 electrons?
The atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons would be 12 amu, which stands for atomic mass units.
What is the atomic mass of an atom that has 6 protons 6 neutrons and 8 electrons?
Carbon-14 contains six protons, six electrons, and eight neutrons per atom. This is the heavyweight form. The atomic mass of carbon is listed as 12.011 atomic mass units per atom.
How can two different elements have the same mass number?
Atoms of an element with the same mass number make up an isotope of the element. Different isotopes of the same element cannot have the same mass number, but isotopes of different elements often do have the same mass number, e.g., carbon-14 (6 protons and 8 neutrons) and nitrogen-14 (7 protons and 7 neutrons).
Can different elements have the same number of neutrons?
Atoms from two different elements may have the same number of neutrons, but never the same number of protons. The number of protons is unique to the element and represents the atomic number.
What is the same for all atoms of the same element?
All atoms of the same element have the same number of protons in their nucleus.



