Which of the following curve is a cooling curve?
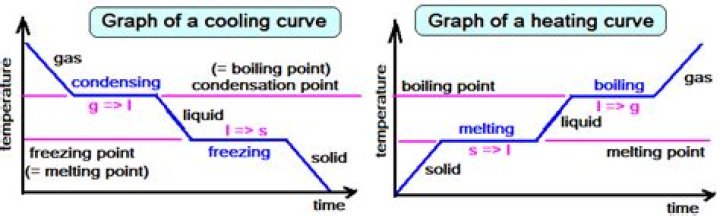
A cooling curve is a line graph that represents the change of phase of matter, typically from a gas to a solid or a liquid to a solid. The independent variable (X-axis) is time and the dependent variable (Y-axis) is temperature. The amount of energy required for a phase change is known as latent heat.
What happens to the particles in a cooling curve?
during melting and evaporating, internal energy increases as the motion of particles increases and bonds are broken. during condensing and freezing, internal energy decreases as the motion of particles decreases and new bonds are formed.
Where are cooling curves used?
A cooling curve is a type of graph used in chemistry, physics, engineering, and other disciplines to chart the progress of a cooling substance.
Is a cooling curve endothermic or EXOthermic?
Cooling curve (EXOthermic physical change):
Temperature decrease: gas (steam) cooling down releases 0.48 calories for each decrease of 1 degrees Celsius for each 1 gram of water.
What does the cooling curve represent Class 11?
The straight horizontal lines in the cooling curve represent the release of latent heat of vaporization (BC) and latent heat of fusion respectively when there is a change of state from gaseous to liquid and liquid to solid.
How do you draw a phase diagram from a cooling curve?
The simplest way to construct a phase diagram is by plotting the temperature of a liquid against time as it cools and turns into a solid. As discussed in Interpretation of cooling curves, the solidus and liquidus can be seen on the graphs as the points where the cooling is retarded by the emission of latent heat.
What is cooling in chemistry?
Cooling is removal of heat, usually resulting in a lower temperature and/or phase change. Temperature lowering achieved by any other means may also be called cooling.
How do you find the cooling curve?
Calculate the cooling rate by dividing each temperature data point by its corresponding time data point then average all of your answers to achieve a cooling rate. In other words, the change in the temperature divided by the change in time will give you an average temperature rate change.
Why is there a plateau in a cooling curve?
Changes of state occur during plateaus because the temperature is constant. The melting and boiling points of the substance can be determined by the horizontal lines or plateaus on the curve. Other substances would of course have melting and boiling points that are different from those of water.
Which is the most accurate statement about the heating curve and energy?
Which is the MOST accurate statement about the heating curve and energy? Your answer: Moving from A to C the temperature and the kinetic energy increases.
Is Cooling linear?
Newton’s law of cooling (generally, black body radiation) is fairly related. However if you’re supplying constant power to a stove heating water, and assuming it’s pretty well insulated so cooling isn’t a problem, the water is gaining a linear amount of energy per time.
What does equilibrium cooling mean?
The liquid and solid phases exist together in equilibrium only at this temperature. When cooled, the temperature of the molten material will steadily decrease until the melting point is reached. Between these two points the liquid and solid phases are in equilibrium.
Which part of a cooling curve shows the changes of state?
Just like heating curves, cooling curves have horizontal flat parts where the state changes from gas to liquid, or from liquid to solid. These are mirror images of the heating curve.
What is cooling curve of paraffin wax?
Paraffin releases the amount of 135.2 j/g of heat energy during the temperature 0 change of 8.9 C. The above DSC curve represents that there is also a small sub cooling during melting and solidification in case of paraffin.
What is the heating and cooling curve of water?
The heating curve for water shows how the temperature of a given quantity of water changes as heat is added at a constant rate. During a phase change, the temperature of the water remains constant, resulting in a plateau on the graph.



