Which of the following best describes atoms the basic unit of matter?
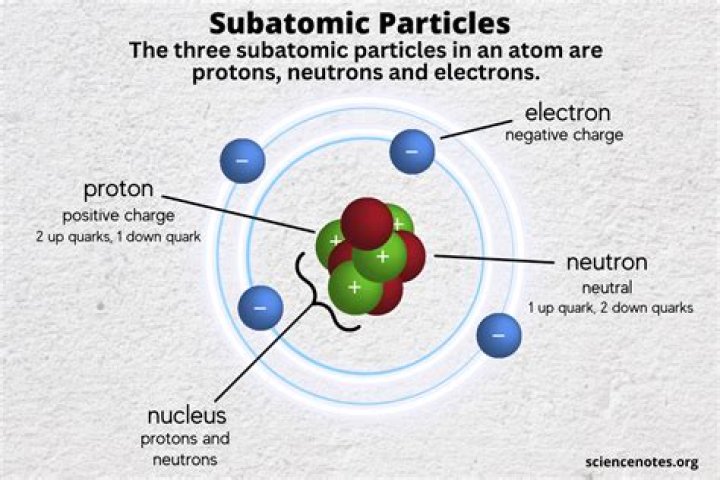
Which of the following best describes atoms, the basic unit of matter? Atoms consist of negatively charged electrons orbiting around positively charged protons and uncharged neutrons.
Which phrase best describes what happens to atoms in a chemical reaction?
In a chemical change, the atoms in the reactants rearrange themselves and bond together differently to form one or more new products with different characteristics than the reactants. When a new substance is formed, the change is called a chemical change.
What phrase best describes a quark?
What phrase best describes a quark? A section of the electron cloud.
Which of the following Cannot be broken down by ordinary chemical means *?
Elements are those pure substances that cannot be decomposed by ordinary chemical means such as heating, electrolysis, or reaction. Gold, silver, and oxygen are examples of elements.
What is atom definition with example?
An atom is any particle of matter at its most basic level which contains at least one proton. Here are some examples of the atoms: hydrogen (H) neon (Ne).
What is atom class 9th?
The smallest tiny particles of matter which can’t be divided further is called atom, i.e., an atom is the smallest building block of matter. For example: Sodium (Na), Hydrogen (H), Oxygen (O), etc.
What scientist said atoms are indivisible?
Leucippus of Miletus (5th century bce) is thought to have originated the atomic philosophy. His famous disciple, Democritus of Abdera, named the building blocks of matter atomos, meaning literally “indivisible,” about 430 bce.
Why do atoms undergo chemical reactions?
Atoms form chemical bonds to make their outer electron shells more stable. The type of chemical bond maximizes the stability of the atoms that form it.
How are atoms held together in molecules quizlet?
The atoms in a compound are held together by chemical bonds. Either “ionic bonds” (formed when one or more electrons are ‘transferred’ from one atom to another) or “covalent bonds” (formed when atoms ‘share’ electrons instead of transferring them).
What best describes a neutral atom?
An atom that has equal number of protons and electrons is called a neutral atom.
What best describes a neutron?
A neutron is a subatomic particle found in the nucleus of every atom except that of simple hydrogen. The particle derives its name from the fact that it has no electrical charge; it is neutral. Neutrons are extremely dense. The number of proton in an element’s nucleus is called the atomic number.
Are atoms made of quarks?
Atoms are constructed of two types of elementary particles: electrons and quarks. Quarks make up protons and neutrons, which, in turn, make up an atom’s nucleus. Each proton and each neutron contains three quarks. A quark is a fast-moving point of energy.
What created atoms?
Atoms were created after the Big Bang 13.7 billion years ago. As the hot, dense new universe cooled, conditions became suitable for quarks and electrons to form. Quarks came together to form protons and neutrons, and these particles combined into nuclei.
Which describe the chemical change caused by heat?
Change of State
If heat is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic. In this instance, heat is increasing the speed of the molecules causing them move faster. If heat is removed from a substance, such as in freezing and condensation, then process is exothermic.
Can pure substances be separated by physical means?
Mixtures can be separated into pure substances by physical methods. Pure substances can be divided into two groups – elements and compounds. Elements are the simplest forms of matter and therefore cannot be broken down into simpler substances by any chemical or physical means.
How do you describe the type of atom?
The number of protons in the nucleus is the atomic number and it defines to which chemical element the atom belongs. For example, any atom that contains 29 protons is copper. The number of neutrons defines the isotope of the element.
What are 4 examples of atoms?
Here are some examples of atoms:
Neon (Ne)Hydrogen (H)Argon (Ar)Iron (Fe)Calcium (Ca)Deuterium, an isotope of hydrogen that has one proton and one neutron.Plutonium (Pu)F-, a fluorine anion.
What is the definition of atom in science?
The atom is considered the basic building block of matter. Anything that has a mass—in other words, anything that occupies space—is composed of atoms. Given that these particles make up atoms, they are often referred to as subatomic particles. There are three subatomic particles: protons, neutrons and electrons.



