What type of bonding is PCl3?
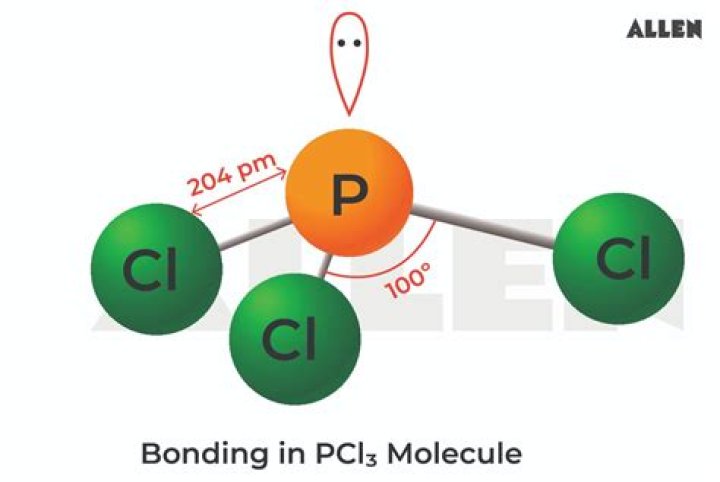
So, this is a polar covalent molecule.
What type of bonding is PCl3?
Phosphorus trichloride (PCl3 ) is a covalent molecule.
Does PCl3 have both ionic and covalent bonds?
The bond between Phosphorus and Chlorine in PCl3 is covalent as chlorine needs one electron and phosphorus has metallic character and easily donate electron.
Why is PCl3 a covalent bond?
PCl3 is a covalent compound as electrons are shared between the P atom and the Cl atoms. P atom is three electrons short of achieving an octet, while the Cl atoms are one electron away. The sharing of electrons results in an inert electron configuration for all the atoms.
Does PCl3 have a nonpolar covalent bond?
The PCl3 molecule is polar in nature due to its asymmetric form and difference in electronegativity. The formal charge on phosphorus of PCl3 molecule is zero. The dipole moment of PCl3 molecule is 0.97 D.
Is LIBR covalent or ionic?
Lithium bromide is an ionic compound of lithium and bromine.
Is PCl3 a dispersion force?
(a) PCl3 is polar while PCl5 is nonpolar. As such, the only intermolecular forces active in PCl5 are induced dipole-induced dipole forces (London dispersion forces). In PCl3, there are also dipole-dipole forces and dipole-induced dipole forces.
Is PCl3 an electronegativity?
The phosphorus atom in the covalent compound phosphorus trichloride, PCl3 , has an electronegativity of 2.19, and the chlorine atoms each have an electronegativity of 3.16, therefore the difference in electronegativity is 0.97, indicating a polar covalent bond between the phosphorus atom and each of the three chlorine
Why is PCl3 a base?
Phosphorus trichloride has a lone pair, and therefore can act as a Lewis base. Remember when you draw the Lewis structure for PCl5 that Phosphorous (P) is in Period 3 on the Periodic table. This means that it can hold more than 8 valence electrons.
Is PCl3 a single double or triple bond?
Phosphorus has one single bond, two double bonds, and one lone pair of electrons. Two of the chlorines have one double bond and two lone pairs of electrons. One chlorine has one single bond and three lone pairs of electrons.
Is PCl3 an electrolyte?
Hence it is a weak Electrolyte. PCl3 is covalent. It undergoes hydrolysis in water to give hydrochloric acid. Which is a strong electrolyte.
Which substance has both ionic and covalent?
Sodium nitrate is a compound with both ionic and covalent bonds.
Is pbcl3 ionic?
Phosphorus trichloride (PCl3 ) is covalent molecule. In the case of phosphorus, 5 covalent bonds are possible – as in PCl5.
Which of the following is nonpolar PCl3?
Due to the electronegativity difference P obtained partial positive charge and Cl obtained partial negative charge i.e.polarity created in PCl3.So,PCl3 is a polar molecule. PCl3 is polar. Chlorine has a higher electronegativity than phosphorus.
Is c2cl2 ionic or covalent?
There is a triple bond between the two carbon atoms and the chlorine and carbon atoms are linked by a single bond. The chlorine atom has six valence electrons on it. it shares one electron each with a carbon atom, and carbon shares its one electron to form a covalent bond.
Is SbF5 polar?
The bond dipole moments in vertical axis cancel out . Similarly the three Sb-F bonds positioned at 120 degrees to each other (top view image ) cancel out each other. Therefore SbF5 in non polar molecule.



