What type of bond is CS2?
The two types of chemical compounds are ionic compounds and molecular (covalent) compounds. Ionic compounds result from ionic bonds where one or more electrons are transferred from one atom to another.
What type of bond is CS2?
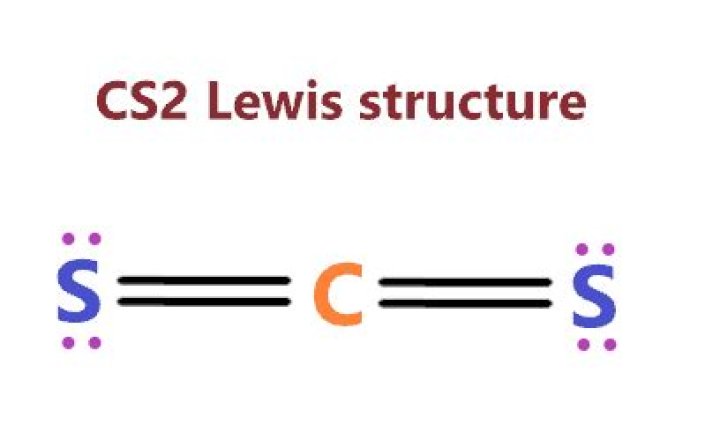
The CS2 molecule is a nonpolar covalent bond with two double bonds. The valence electrons that form this type of bond are in 2s and 2p orbitals of the carbon atom.
Is co2 ionic or molecule?
Carbon and oxygen are non-metals, thus we know carbon dioxide is a covalent compound.
Is carbon disulfide CS2 ionic or molecular?
Answer: CS2 ( Carbon disulfide ) is a covalent bond What is chemical bond, ionic bond, covalent bond? Top Answer. A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms.
Is CS2 a polar molecule?
Is CS2 polar or nonpolar? – Quora. Carbon disulfide is a non-polar molecule. Polarity in relatively simple terms arises as a result of uneven sharing of bond electrons in a covalent molecule. The cause of this is differences in electronegativities of the elements making up the molecule.
What is a molecular compound?
A molecular compound consists of molecules whose formula represents the actual number of atoms bonded together in the molecule. The atoms are joined to give a definite shape which is defined by the angles between the bonds and by the bond lengths. Some examples are shown below. H.
What type of compound is carbon disulfide?
Carbon disulfide, also known as CS2, belongs to the class of inorganic compounds known as other non-metal sulfides. These are inorganic compounds containing a sulfur atom of an oxidation state of -2, in which the heaviest atom bonded to the oxygen belongs to the class of other non-metals.
Is Cao ionic or covalent?
Calcium oxide is ionic because it is formed between a metal and non-metal and bonds formed between a metal and non-metal atoms are ionic. A bond formed between nonmetal atoms is covalent.
Is CO2 molecular?
Carbon dioxide is a molecule with the molecular formula CO2. Carbon dioxide, CO2, is a colorless gas. It is made of two oxygen atoms covalently bonded to one carbon atom.
Is CO2 molecular compound?
CO2 is a molecular compound. Ionic compounds are composed of a non-metal and a metal element. Molecular compounds are made up of two non-metals.
Is Co a molecular covalent compound?
The carbon monoxide molecule is correctly represented by a triple covalent bond between the carbon and oxygen atoms. One of the bonds is a coordinate covalent bond, a covalent bond in which one of the atoms contributes both of the electrons in the shared pair.
What is the compound CS2?
carbon disulfide (CS2), also called Carbon Bisulfide, a colourless, toxic, highly volatile and flammable liquid chemical compound, large amounts of which are used in the manufacture of viscose rayon, cellophane, and carbon tetrachloride; smaller quantities are employed in solvent extraction processes or converted into
Is carbon a disulfide molecule?
Only molecules of carbon disulfide have the properties of carbon disulfide, and it is made of molecules.
Is NH4NO3 a covalent compound?
Ionic bond. NH4NO3 is a nitrate salt of the ammonium cation. Since ammonium is a cation and bonds with the anion nitrate, hence the compound is bonded by an ionic bond.



