What shape is ICl3?
Geometry of ICl3 is tetrahedral.
Is ICl3 planar?
Since it is single-bonded with three chlorine atoms, there are no lone pairs. Since it is single-bonded with three chlorine atoms, there are no lone pairs. So, the steric number is 3+0=3. Hence it is of the molecule type $A{{X}_{3}}{{E}_{0}}$ and has a trigonal planar shape where all bond angles are $120{}^circ $.
What is a trigonal shape?
Trigonal planar is a molecular shape that results when there are three bonds and no lone pairs around the central atom in the molecule. The pairs are arranged along the central atom’s equator, with 120° angles between them.
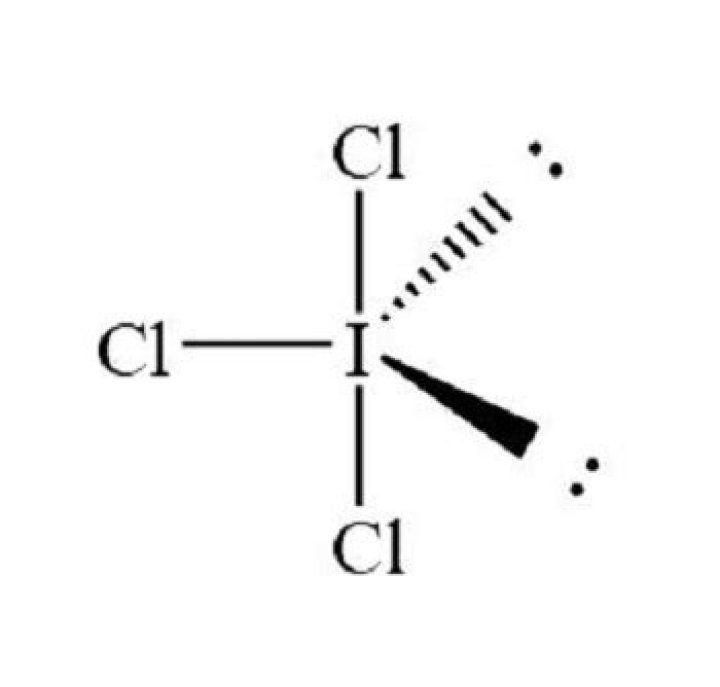
The geometry of ICl3 is trigonal bipyramidal with a T-shaped molecular shape. ICl3 has three bond pairs and two lone pairs of electrons. The hybridization of the iodine atom in ICl3 is sp3d with trigonal bipyramidal geometry.
Why is ICl3 not trigonal planar?
The reason why ICl3 exists in T-shaped geometry, with all of its angles at slightly less than 90 degrees, is because it is the most stable in that structure.
ICl3 is made from only nonmetals and is a covalent compound.
How do you write the name ICl3?
Iodine trichloride | ICl3 – PubChem.
Is NH3 planar?
NH3 is not planar, BF3 is planar.
The Lewis structure of ammonia, NH3 , would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. This is the reason why ammonia acts as a Lewis base, as it can donate those electrons.
What is the geometry of the NO2 anion?
We can see that NO2 has a bent molecular geometry and the angle is around 120 degrees. But here we have some exceptions. In NO2, we have 2 Bond Pairs and 1 lone electron. If we look at the nitrite ion NO2-, we have 2 Bond Pairs and 1 Lone pair of electrons.
What is the geometry of brf3?
BrF3 molecular geometry is said to be T-shaped or Trigonal Bipyramidal with a bond angle of 86.2o which is slightly smaller than the usual 90°. This angle formed due to the repulsion generated by the electron pairs which is greater than that of the Br-F bonds.
What is the molecular structure of NCl3?
The molecular geometry of NCl3 is trigonal pyramidal and its electron geometry is tetrahedral. NCl3 lewis dot structure contains 1 lone pair and 3 bonded pairs. The net dipole moment of Nitrogen trichloride is 0.6 D.
why ICl3 get dimerised.
As electronegativity decreases bond angle increases . We know bond angle in IF3 is less than 90o . But as we move from IF3 to ICl3 bond angle between lone pairs and bond pairs increases and approaches 90o. Therefore ICl.
What is the formal charge on the central atom in a molecule of ICl3?
The number of bonding electrons (BE) of iodine is 6. Hence, the formal charge on iodine atom is 0.
What makes a trigonal pyramidal?
Central atom will at the apex and three other atoms or ligands will be at one base, where they are in the three corners of a triangle. There is one lone pair of electrons in the central atom. So when the position of the lone pair is neglected, the remaining bonds make the trigonal pyramidal geometry.
Is T shaped polar?
No known compounds do this however, so the only “real” T-shaped molecules have trigonal bipyramid electroni geometries and not octahedral. POLARITY: NON-POLAR – If this did exist, the whole molecule has a perfect symmetry and therefore non-polar (assuming equivalent atoms in the two peripheral positions).



