What is the valence charge of Aluminium?
Aluminium has 3 valence electrons.
What is the valence charge of aluminum?
The valence shell of aluminum has three electrons, and per the octet rule, these three electrons are lost resulting in just 10 electrons and 13 protons. Aluminum then has three excess protons so the charge of a base aluminum ion is 3+. That’s the quick answer regarding how aluminum’s charge is 3+.
What is the charge of aluminium?
The charge of an aluminum ion is typically 3+. This is because the element’s atomic number is 13, reflecting the fact that it has 13 electrons and 13 protons. The valence shell of aluminum has three electrons, and per the octet rule, these three electrons are lost resulting in just 10 electrons and 13 protons.
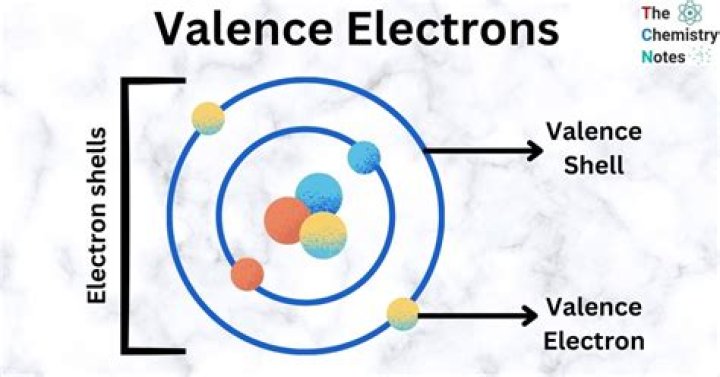
Just by looking at the electron configuration, you see that there are three valence electrons.
Why valency of aluminum is 3?
Answer: Atomic number of aluminium is 13 so its electronic configuration is 2, 8, 3. Since it has 3 electrons in its valence shell, it will lose 3 electrons to complete its octet hence its valency is +3.
For neutral atoms, the number of valence electrons is equal to the atom’s main group number. The main group number for an element can be found from its column on the periodic table. For example, carbon is in group 4 and has 4 valence electrons. Oxygen is in group 6 and has 6 valence electrons.
Is aluminum negatively charged?
It is also important to note that the aluminum can is not a charged object. While it contains charges, it does not have an overall charge. The number of positive and negative charges is equal. This balance of the two types of charges is evidence that the aluminum can is neutral.
What is the ion formula of aluminum?
Al+3 is “aluminum ion”.
Aluminium-ion batteries are a class of rechargeable battery in which aluminium ions provide energy by flowing from the positive electrode of the battery, the anode, to the negative electrode, the cathode. When recharging, aluminium ions return to the negative electrode, and can exchange three electrons per ion.
Does aluminum have 13 valence electrons?
Aluminum atoms contain 13 electrons and 13 protons. There are 3 valence electrons in the outer shell. In standard conditions aluminum is a fairly soft, strong, and lightweight metal. Pure aluminum is a very reactive element and is rarely found on Earth in its free form.
What is the electron number of aluminium?
An aluminium atom has 13 electrons, arranged in an electron configuration of [Ne] 3s2 3p1, with three electrons beyond a stable noble gas configuration.
How many neutrons does aluminium have?
The number of protons in Aluminium is 13. The 27 means the atomic mass is 27 . The number of protons plus the number of neutrons is 27. That means the number of neutrons is 27–13=14.
– Valency is the number of electrons required by an atom to complete its octet to attain the nearest noble gas configuration and become stable. So, the valency of Aluminum is 5.
Does Aluminium have variable valency?
We can define variable valency as a phenomenon that is generally found in transition elements. In iron (II) oxide, the oxidation state/valency of iron is +2 and in iron (III) oxide, the oxidation state/valency of iron is +3. We can term as variable valency. Aluminum does not belong to the transition element.
How many electrons are in the valence shell of aluminum?
This is a total of ten electrons in filled shells. That leaves 3 electrons in the third shell. This is the valance shell since it is not filled. So the 3s23p1 electrons are the valance electrons for a total of 3 valance electrons.
How many core and valence electrons does aluminum have?
So for the element of ALUMINUM, you already know that the atomic number tells you the number of electrons. That means there are 13 electrons in a aluminum atom. Looking at the picture, you can see there are two electrons in shell one, eight in shell two, and three in shell three.



