What is the total number of atoms in the formula Al2 CO3 3?
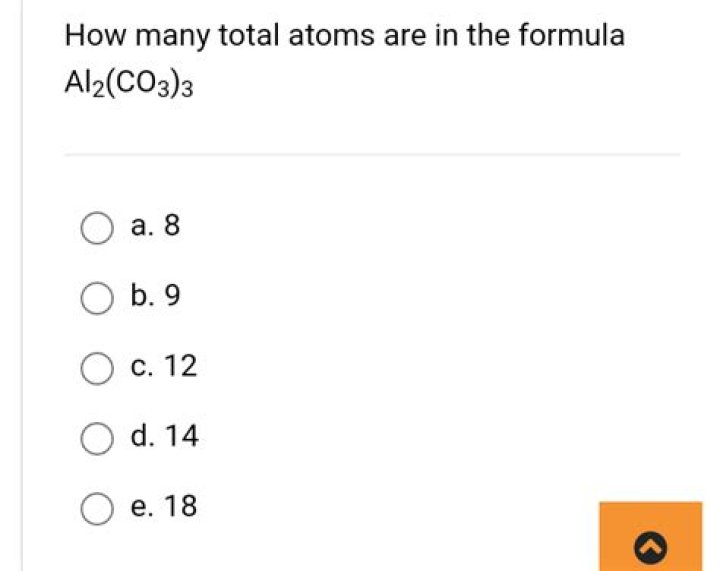
Explanation: And so there are 2×Al+3×C+9×O=14 atoms .
How many atoms are there in Al2 SO4 3?
Answer: In this case, Al2 means 2 aluminums, (SO4)3 means 3 (SO4)s. The parentheses mean that the 3 has to be multiplied by whatever number of atoms you have of each element inside: 3×1 sulfurs, 3×4 oxygens. Now for the grand tally: 2 aluminum atoms + 3 sulfur atoms + 12 oxygen atoms = 17 atoms total in each molecule.
How many atoms are there in Aluminium?
Finally, to convert the number of moles to atoms, use the fact that 1 mole of aluminium must contain 6.022⋅1023 atoms of aluminium → this is known as Avogadro’s constant.
How many atoms is 5.80 moles?
5.80 mol He 6.02210^23 atoms He/ 1 mol He. This cancels out moles, leaving 3.49*10^24 atoms He.
How many total atoms are there in one molecule of aluminum sulfate?
-Now, we have two aluminium atoms, three sulphur atoms and twelve oxygen atoms in one molecule of aluminium sulphate. 2+3+12=17 , i.e. seventeen atoms.
How many atoms are in caoh2?
Calcium hydroxide, Ca(OH) 2 consists of one calcium atom (A r = 40), two oxygen atoms (A r = 16) and two hydrogen atoms (A r = 1).
How many atoms are in each element?
An atom is an element. The two words are synonymous, so if you’re looking for the number of atoms in an element, the answer is always one, and only one.
What are number of atoms?
atomic number, the number of a chemical element in the periodic system, whereby the elements are arranged in order of increasing number of protons in the nucleus. Accordingly, the number of protons, which is always equal to the number of electrons in the neutral atom, is also the atomic number.
What is the formula for the number of atoms?
The equation is fairly simple. The number of atoms of ANY substance in a volume is: # of atoms = N * (density) * volume / (Molecular Weight). N is a constant called Avogadro’s number and its equal to 6.022*1023 atoms/mole.
How many moles of atoms are in a mole of Al2O3?
1 mole Al2O3 contains 2 moles of Al and 3 moles of O. Weight of one mole of oxygen is 16 g and Al is 27 g. So moles of O are 3 and Al are 2.
How many atoms does C12H22O11?
A molecule of sucrose (C12H22O11) has 12 carbon atoms, 22 hydrogen atoms and 11 oxygen atoms.
How much is an atom of aluminum?
The cost of one atom of aluminum is $2.06E-25.
How many atoms are in 4 moles of aluminum?
Therefore, [2.41times 10{}^text{2}] atoms/mol of Aluminum are present in $4$ mol of Aluminum. Additional Information:Why is Avogadro’s number referred to as a mole?
How many atoms are contained in 135g Al?
SOLVED:A mass of 135 g of a certain element is known to contain 30.1 times 10^{23} atoms.



