What is the shape of NO2 negative?
We can see that NO2 has a bent molecular geometry and the angle is around 120 degrees.
Is NO2 linear or bent?
NO2 is a bent molecule; however, when you remove an electron from it, making it NO2+, the molecule becomes linear due to the loss of a lone electron. On the other hand, nitrogen dioxide, NO2, is an AX2E species, and it has an angle of 134 degrees. The additional lone pair on the SF2 molecule makes the angle smaller.
Is NO2 negative linear?
NO2 is a bent molecule; however, when you remove an electron from it, making it NO2+, the molecule becomes linear due to the loss of a lone electron. In NO2+, there is no repulsion taking place between the two O atoms and the lone electron on the central atom.
$N{O_2}$ has two bond pairs and one lone pair, so it will have triangular planar geometry and $s{p^2}$ hybridization and $NO_2^ – $ will have two bond pairs, one lone pair, and also a negative charge.
Is NO2 trigonal planar?
In this example, NO2, the Lewis diagram shows nitrogen at the center with one lone electron. Hence the molecule has three electron pairs and is trigonal planar for electron pair geometry.
However, the shape of the molecule is described in terms of the positions of the atoms, not the lone electron pairs. Thus, NO2– is said to have a bent shape, not a trigonal planar one. It is assumed that the lone pair is “invisible” when describing the shape of the molecular geometry.
Is BrO3 trigonal planar?
The total valence electron available for drawing the Bromate ion (BrO3-) lewis structure is 26. The hybridization in BrO3- is Sp3. Total 16 lone pairs electrons and 10 bonded pairs electrons present in BrO3- lewis structure. The molecular geometry of BrO3- is trigonal pyramidal and electron geometry is tetrahedral.
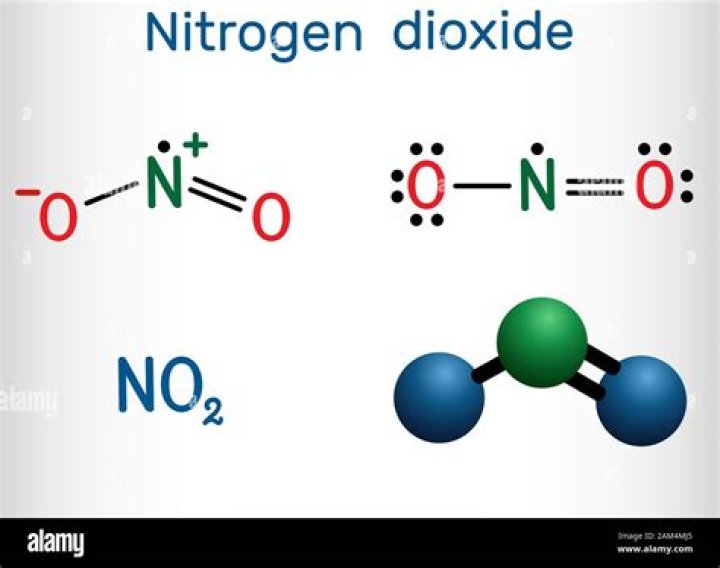
Is NO2 a structure?
Nitrogen Dioxide (NO2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. At room temperatures, nitrogen dioxide is a reddish-brown gas that has a density of 1.8 g/dm3.
Although the shape of the N2O molecule is linear, the sharing of electrons between the atoms is unequal.
Why is NO2 sp2?
When the bonding takes place, the two atoms of oxygen will form a single and a double bond with the nitrogen atom. Meanwhile, nitrogen must have three hybridized orbitals that will be used to harbour two sigma bonds and one electron. This results in sp2 hybridization.
Is n2o linear or nonlinear?
2 Answers. Nitrous oxide is linear.
What is the Lewis structure of NO2 negative?
After determining how many valence electrons there are in NO2-, place them around the central atom to complete the octets. There are a total of 18 valence electrons for the Lewis structure for NO2-. Nitrogen is the least electronegative atom in the NO2- lewis structure and therefore goes in the center of the structure.
| NO2 Acidity. Nitrogen dioxide is an acidic gas and produce an acidic solution in the water (mixture of acids). Otherwise, we can say, NO2 is one of the strong acidic gas in chemistry.



