What is the shape and polarity of CH2Cl2?
The CH2Cl2 molecule has a tetrahedral geometry shape because it contains two chlorine and two hydrogen atoms. There are two C-H and two C-Cl bonds at the CH2Cl2 molecular geometry. After linking the two hydrogens and two chlorine atoms in the tetrahedral form, it maintains the tetrahedral-like structure.
What is the shape and polarity of CH2Cl2?
Firstly, it has polar molecules shaped trigonal pyramidal. The second reason is that the geometry of the compound is tetrahedral, which according to chemistry, has a central carbon atom surrounded by four other atoms. The third is the electronegativity between C-H and C-Cl is 0.4 and 0.6, respectively.
Is CH2Cl2 trigonal planar?
If it were planar, then there would be isomers of CH2Cl2 wherein the chlorine atoms would be cis or trans to each other. However, CH2Cl2 always crystallizes as a single substance, so there are no isomers, and the only possibility left is tetrahedral.
Is CH2Cl2 asymmetric or symmetrical?
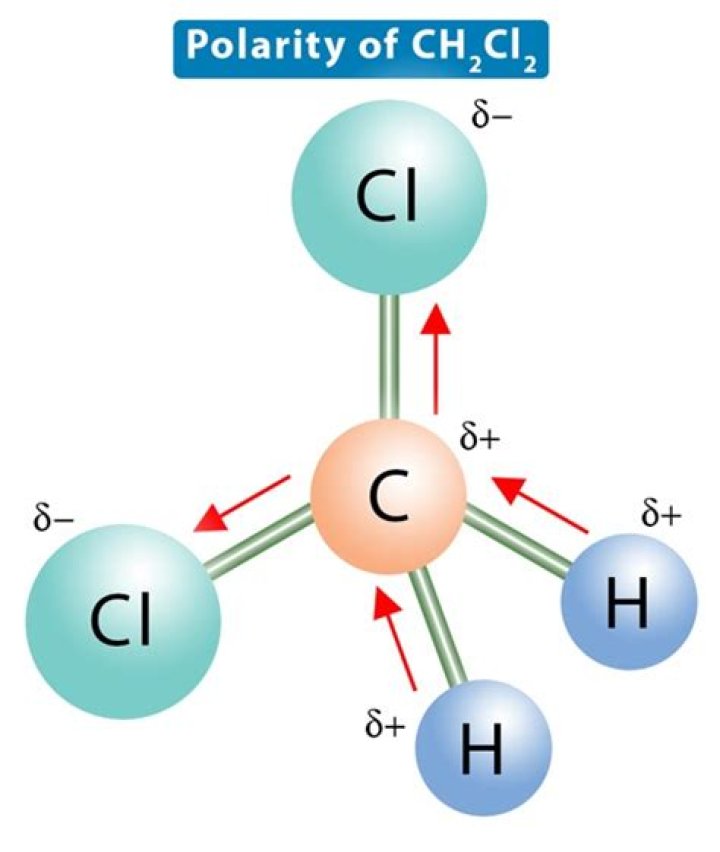
Although the bond arrangement around the C atom in CH2Cl2 is symmetrical, the differing polarities of the C–H and C–Cl bonds means the effect of the polar bonds is not cancelled, so the molecule is polar.
What would the polarity of a square planar CH2Cl2?
Originally Answered: Is CH2Cl2 polar or nonpolar? It’s polar as its geometry is tetrahedral. As chlorine is more electronegative than carbon dipole moment is observed towards chlorine. Due to tetrahedral shape the vector sum also will be effective and it will show polarity.
What is the point group of CH2Cl2?
CH2Cl2 1)belongs to point group D2 2)has one plane of symmetry, sigma h 3)has one C2 axis of rotation | Study.com.
Which is the best description of the molecule dichloromethane CH2Cl2 )?
Dichloromethane appears as a colorless liquid with a sweet, penetrating, ether-like odor. Noncombustible by if exposed to high temperatures may emit toxic chloride fumes.
Why is CCl2H2 polar?
Re: Polarity of CCl2H2
Since H and Cl have different electronegativities, the C-H and C-Cl dipole moments are also different. So although the 4 dipole moments do point in the same direction, they are not equivalent and thus do not cancel, making the overall molecule polar.
Is CH2Cl2 tetrahedral polar or nonpolar?
Because of its tetrahedral geometrical structure and the disparity in electronegativity of the Carbon, Hydrogen, and Chlorine atoms, CH2Cl2 is a polar molecule.
Is dichloromethane more polar than water?
Yeh. CH2Cl2 is barely polar actually since C-Cl has deltaEN=0.5 where C-H gives a 0.4. Much less polar than water for sure..
Which statement best describes the polarity of CH2Cl2?
Which statement best describes the polarity of CH2Cl2? The molecule is always polar.
What is the direction of dipole moment in CH2Cl2?
Dipole-Dipole Forces
In contrast, dichloromethane (methylene chloride, CH2Cl2) is a polar molecule with a net polarity away from the partially positive carbon atom toward the partially negative chlorine atoms.



