what is the product of the hydrogenation of an alkene?, check these out | What is the product of the hydrogenation?
The product is an alkane. Hydrogenation is used in the food industry to convert liquid oils into saturated fats. This process yields semi-solid products like shortening and margarine.
What is the product of the hydrogenation?
The most commonly hydrogenated product is vegetable oil. Hydrogenation converts vegetable oil from a liquid to a solid or semi-solid fat. D-Sorbitol syrup is made by hydrolysis of starches to make dextrose, the dextrose is then hydrogenated to create sorbitol or sugar alcohol.
What is hydrogenation reaction of alkenes?
When alkenes react with hydrogen gas in the presence of a variety of metal catalysts, a hydrogen molecule will be added to the double bond in the way that each carbon atom bonded with one hydrogen atom, such addition reaction is called hydrogenation.
What is the product of the hydrogenation of an alkyne?
Mechanism of the Hydrogenation
When an alkyne is hydrogenated with Lindlar’s catalyst, we get an alkene. What’s important to realize is that we always get an alkene in which the hydrogen atoms are on the same side of the carbon-carbon double bond.
What is hydrogenation 10th?
Answer: The addition of hydrogen to an unsaturated hydrocarbon to get a saturated hydrocarbon in presence of nickel or palladium as catalyst is called hydrogenation.
Which solvents provide better alkene hydrogenation activity?
Alcohols (e.g., methanol or ethanol) are the most widely used solvents for reported heterogeneous catalytic hydrogenation reactions [21]. Wei et al. [22] have shown that Pd catalysts exhibit higher activity for hydrogenation reactions in toluene and ethanol, than in DMF, acetonitrile, or water.
What is the product produced from the halogenation of alkenes?
The halides add to neighboring carbons from opposite faces of the molecule. The resulting product is a vicinal (neighboring) dihalide.
What is the reagent for hydrogenation of an alkene?
The reaction is also known as the hydrogenation of alkene. The reaction occurs in the presence of a finely divided metal catalyst such as nickel (Ni), palladium (Pd), platinum (Pt), or rhodium (Rh) at a temperature of about 150°C.
What happens during an alkene addition reaction?
The most common type of reaction for alkene is the addition reaction to C=C double bond. In addition reaction, a small molecule is added to multiple bond and one π bond is converted to two σ bonds (unsaturation degree decreases) as a result of addition. Addition reaction is the opposite process to elimination.
What type of reactions do alkenes undergo?
Alkenes undergo addition reactions, adding such substances as hydrogen, bromine, and water across the carbon-to-carbon double bond.
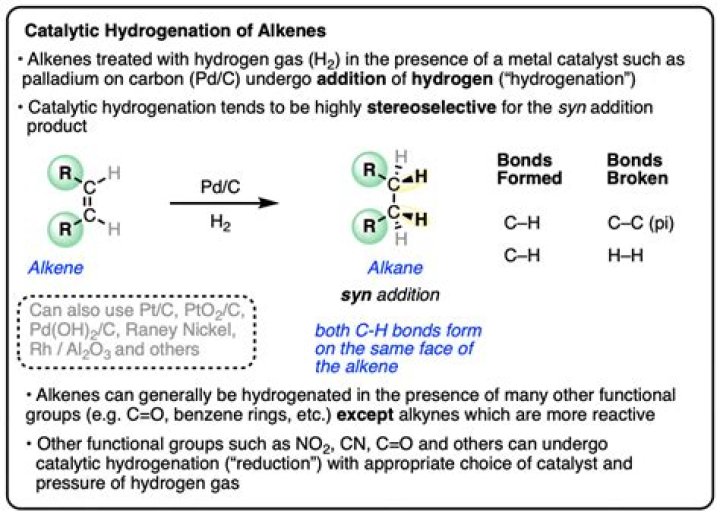
What is catalytic hydrogenation of alkenes?
Catalytic hydrogenation is hydrogenation in presence of catalysts. Addition of hydrogen to alkenes is an exothermic (releasing heat energy) reaction, requiring the use of a transition metal catalyst due to the high energy barriers to direct the reaction between alkenes and hydrogen gas.
What are the characteristic reactions of alkenes and alkynes?
Addition reactions involving alkenes and alkynes include hydrogenation, halogenation, and hydrohalogenation. Alkenes and alkynes are useful reagents in polymer synthesis—an important industrial application.
What is the chemical properties of alkene?
Alkenes are non-polar, and they are both immiscible in water and less dense than water. They are generally soluble in organic solvents. In addition, they do not conduct electricity.
How are the following compounds obtained from alkene?
When a stream of ozonised oxygen is passed through a solution of an alkene, in organic solvent, an unstable addition cyclocompound, ozonide is formed which on reduction with zinc dust and water forms an aldehyde or a ketone or a mixture of both.
What is the product in the hydration of alkene?
An alkene does not react with pure water, since water is not acidic enough to allow the hydrogen to act as an electrophile to start a reaction. However, with the presence of small amount of an acid, the reaction does occur with a water molecule added to the double bond of alkene, and the product is an alcohol.
What is hydrogenation in Class 10 which chapter?
Chapter 4: Carbon and Its Compounds
Answer: The addition reaction is a type of reaction in which the unsaturated hydrocarbon is converted into saturated hydrocarbon by reacting with hydrogen gas in presence of nickel. This reaction is also called hydrogenation reaction.
What is the process of hydrogenation?
Lesson Summary. Hydrogenation is the process where hydrogen atoms bind to the double bond of a compound, facilitating its conversion to a single bond, in the presence of a catalyst. Hydrocarbons with double bonds are classified as unsaturated.
What is hydrogenation reaction give an example?
Hydrogenation Is a chemical reaction between hydrogen(h2) and a compound or element in presence of a catalyst. Example: conversation of ethene to ethane.



