What is the monomer of Polychloroethene?
Poly(chloroethene), usually known as polyvinyl chloride or just PVC, is the most versatile plastic and, after poly(ethene), the most widely used. The variety of uses comes from its ability to be formulated with different additives and with different molecular masses, giving plastics that range from rigid to pliable.
What is the monomer of Polychloroethene?
The old name for poly(chloroethene) was polyvinyl chloride, hence the use of the commercial name PVC, and is made from polymerising the monomer chloroethene, CH2=CHCl, (old name vinyl chloride).
What a polymer is?
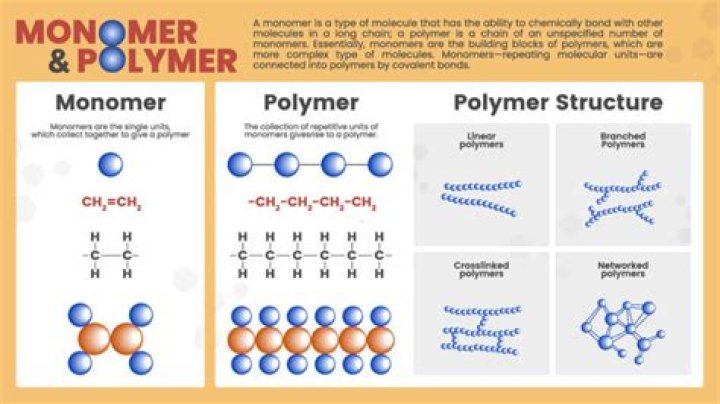
A polymer is any of a class of natural or synthetic substances composed of very large molecules, called macromolecules, which are multiples of simpler chemical units called monomers. Polymers make up many of the materials in living organisms and are the basis of many minerals and man-made materials.
What is poly chloroethene made from?
Poly(chloroethene) is commonly known by the initials of its old name, PVC. Poly(chloroethene) is made by polymerising chloroethene, CH2=CHCl. Working out its structure is no different from working out the structure of poly(propene) (see above).
What are examples of polymers?
Examples of synthetic polymers include nylon, polyethylene, polyester, Teflon, and epoxy. Natural polymers occur in nature and can be extracted. They are often water-based. Examples of naturally occurring polymers are silk, wool, DNA, cellulose and proteins.
How is Polychloroethene made?
PVC is made by free-radical polymerization in suspension. The monomer (bp 259 K) is polymerized in aqueous dispersion at 325-350 K. Pressure (13 atm) is used to keep the monomer in a liquid phase. For polymerization to be controlled, an initiator is needed.
How do polymers work?
A polymer is a large molecule made up of smaller, joined-together molecules called monomers. Monomers join together to make polymer chains by forming covalent bonds—that is, by sharing electrons. Other bonds then hold the groups of chains together to form a polymer material.
Is polymer A plastic?
Plastics are a group of materials, either synthetic or naturally occurring, that may be shaped when soft and then hardened to retain the given shape. Plastics are polymers. A polymer can be thought of as a chain in which each link is the “mer,” or monomer (single unit).
What are polymers used for?
Uses of polymers
Polymers are used in almost every area of modern living. Grocery bags, soda and water bottles, textile fibers, phones, computers, food packaging, auto parts, and toys all contain polymers. Even more-sophisticated technology uses polymers.
Is poly chloroethene an addition polymer?
Modelling addition polymerisation
The polymer, poly(chloroethene) is also known as poly(vinyl chloride), or PVC. The diagram shows the structure of propene.
Can alkanes be polymerised?
In chemical compounds, polymerization can occur via a variety of reaction mechanisms that vary in complexity due to the functional groups present in the reactants and their inherent steric effects. Alkanes can also be polymerized, but only with the help of strong acids.
What is the difference between ethene and polyethylene?
is that polyethylene is (organic compound) a polymer consisting of many ethylene monomers bonded together; used for kitchenware, containers etc while ethene is (organic compound) the official iupac name for the organic chemical compound ethylene the simplest alkene, a colorless gaseous (at room temperature and pressure
What are some polymers used in everyday life?
Let’s take a look at some of the most commonly used polymers in everyday life.
Polyethylene.Polyester.Acrylics.Polyvinyl Chloride (PVC)Polypropylene (PP)Rubber.Teflon.Nylon.
How do you explain polymers to a child?
Polymers are very big molecules made up of many smaller molecules layered together in a repeating pattern. In fact, the word polymer is Greek for ‘many parts. ‘ The smaller molecules that come together to form polymers are called monomers–small units that link together over and over to form a large polymer.
What are the 3 main types of polymers?
There are 3 principal classes of polymers – thermoplastics, thermosets, and elastomers. Differentiation between these classes is best defined by their behaviour under applied heat. Thermoplastic polymers can be either amorphous or crystalline. They behave in a relatively ductile manner but often have low strength.
Why are brackets used in polymers?
4Name one use of a polymer. A polymer can often be made from many monomers of the same type joined together. The displayed formula of polymers are written as repeating units with a square bracket at each end. A small n follows the last bracket to show that there could be more repeating units and the chain could go on.
How do alkenes Polymerise?
During the addition polymerization process, the pi bonds of the double bonds in each alkene molecule sort of break open, thereby allowing the free bonds to join with one another to form a chain known as poly(alkene) or polythene. This chain contains units that repeat themselves, known as repeating units.
How do alkenes form polymers?
Pressure and heat energy open the double bonds in the alkene and make it bond to form large polymer molecules.



