What is the molecular geometry of IF4 -?
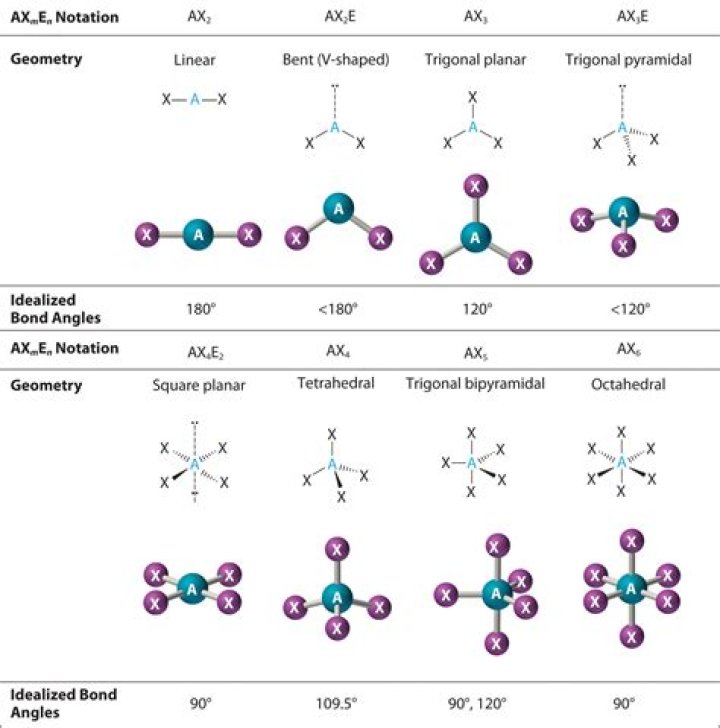
So Hybridization = [s{p^3}{d^2}], the shape of the molecule is octahedral geometry, but the geometry of the atom takes a square planar shape is due to iodine carries two lone pairs of electrons, one above the plane and one below the plane hence the shape of the molecule is square planar.
What is the bond angle of ch2ch2?
Ethene. The simplest alkene ethene (H2C=CH2) is planar with H-C-H and H- C-C bond angles that are close to 120°. Figure 8.2 These 120° bond angles and the planar geometry are consistent with sp2 hybridization for each of ethene’s C atoms (Chapter 1).
Which bond angle is smallest?
Water has the smallest bond angle.
What is the value of the bond angles in BCl3?
All the three bond angles in boron trichloride (BCl3 B C l 3 ) are equal and equal to 120°.
Is IF4 a bond angle?
Its coordination number, which is given by the number of atoms that are bonded to iodine, is equal to 4. For a square planar molecule, ideal bond angles measure 90∘ , so you can predict that the IF4− ion will have, for all intended purposes, bond angles of 90∘ .
What is the hybridization of I in IF4+?
IF4+ is sp3d hybridized; its electron domain geometry is trigonal bipyramidal; and its molecular geometry is seesaw.
What is the bond angle in CH3?
CH3- is trigonal pyramidal with bond angles of slightly less than 109.5 degrees because of the lone pair, like the other molecules you mentioned.
What is the bond angle of HNH?
(The empirical H-N-H bond angles in ammonia are 107.8o.)
Which of the following is smallest angle?
The smallest angle is 1. It is smaller than a right angle, so we call this an acute angle. The largest angle is 2.
What is the value of the bond angle in ICl2 − ICl2 −?
The bond angle in ICl2- molecule is 180º.
What is the value of the bond angle in XeCl2 XECL 2?
180° is the value of the bond angle in XeCl2.
What is the value of the bond angle in XeCl2 enter the bond angle of the molecule?
What is the value of the bond angle in XeCl2? XeCl2 has 8 + 2(7) = 22 valence electrons. a linear molecular structure with a 180° bond angle.



