What is the molecular geometry of ch3+?
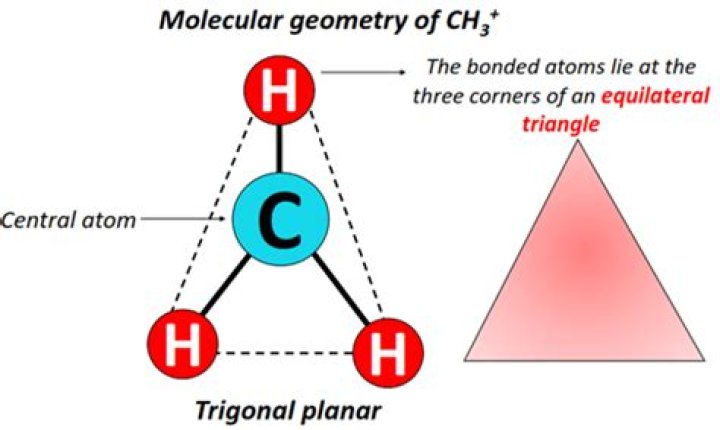
The carbon atom in the ion CH+3 is sp2 hybridized carbon and therefore, the geometry around this atom is trigonal planar. In this case, the H−C−H bond angle is 120∘ . However, the carbon atom in CH4 is sp3 hybridized carbon and therefore, the geometry around this atom is tetrahedral.
Is I3 linear?
I3- molecular geometry is linear. While there are three Iodine atoms, one of the atoms has a negative charge which further gives 3 lone pairs of electrons and 2 bond pairs. Its steric number will be 5. The three lone pairs will repel each other and take up equatorial positions.
What is the molecular geometry of CF4?
10.36 (a) CF4: Electron geometry-tetrahedral; molecular geometry-tetrahedral; bond angle = 109.5° Draw a Lewis structure for the molecule: has 32 valence electrons.
Is ClF4+ polar or nonpolar?
So this molecule is polar.
Does CH3+ have trigonal planar shape?
Explanation: The carbon atom in the ion CH3+ is sp2 hybridized carbon and therefore, the geometry around this atom is trigonal planar.
Are CH3+ and CH3 polar?
CH3+ is nonpolar, because even though C has a higher electronegative charge, the force is being pulled/shared equally between the 3 Hydrogen atoms in a trigonal planar molecular shape.
Is I3 trigonal?
I3- has 2 bonds and 3 lone pairs for 5 regions of electron density. Placing that into a trigonal bipyramidal structure, the lone pairs go equitorial and the two other I atoms go axial. With the three atoms in a line, the molecule is linear.
Why is I3 polar?
I3- is formed by the reaction of I2 (iodine) with I- (iodide). When you add Iodine crystals to a solution of potassium iodide in water, you will generate a fair amount of KI3 (potassium triiodide). I3- is most definitely polar, since it is an anion and carries a negative charge.
Why is I3 not bent?
Since I3+ has sp3 hybridisation and two lone pair on central I atom therefore its shape would be Bent or V shape. Since I3+ has sp3 hybridisation and two lone pair on central I atom therefore its shape would be Bent or V shape. The tri-iodide ion has a total of 9 lone pairs.
What is the the shape molecular geometry of BeBr2?
The BeBr2 molecule has a linear shape because it contains two bromine atoms. There are two Be-Br bonds at the linear BeBr2 molecular geometry. After linking the two bromines in the linear form, it maintains the linear-like structure.
What is the molecular geometry of if 5?
IF5 contains five bonded and one nonbonded electron domains, giving an octahedral e- domain geometry and a square pyramidal molecular geometry (AX5E). The bond angles are compressed relative to those in a perfect octahedron due to the lone pair spreading out more in space than bonded pairs.
Is cif3 polar or nonpolar?
ClF3 Polar or Nonpolar
ClF3 is a polar molecule due to its asymmetrical structure and the presence of two lone pair electrons, which results in an unequal distribution of charge and so makes it polar.



