What is the mass in grams of SiO2?
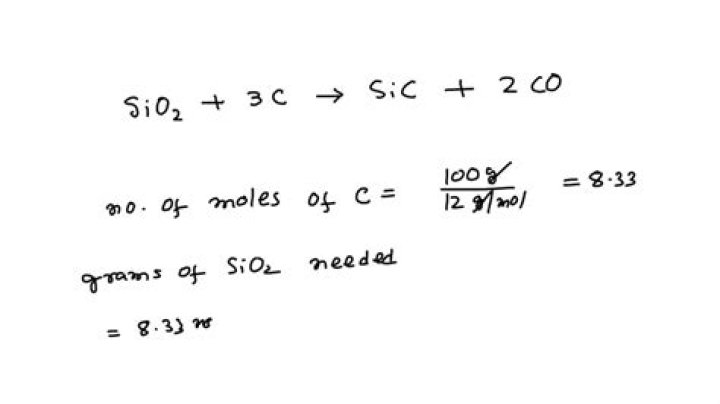
The inorganic compound comprising two oxygen and silicon atom is silicon dioxide with the molecular formula SiO2 S i O 2 . The molar mass for this compound is 60.08 g/mol. It has an amorphous transparent solid in appearance with whitish-yellow color.
How many grams are in one mole of SiO2?
Explanation: And what is the mass of a mole of silicon atoms? It is 28.1⋅g .. And thus SiO2 has a molar mass of (28.1+2×16.0)⋅g⋅mol−1 .
How many moles of Si are in SiO2?
The formula tells us the proportions of the things in a compound. In this case, each mole of SiO2 is made up of 1 mole of Si and 2 mole of O .
How do I find molar mass?
Multiply the atomic weight (from the periodic table) of each element by the number of atoms of that element present in the compound. 3. Add it all together and put units of grams/mole after the number. For many (but not all) problems, you can simply round the atomic weights and the molar mass to the nearest 0.1 g/mole.
How many grams of SiO2 are present in 0.8 mole?
We assume you are converting between moles SiO2 and gram. You can view more details on each measurement unit: molecular weight of SiO2 or grams This compound is also known as Silicon Dioxide. The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles SiO2, or 60.0843 grams.
How many molecules are in 0.0125 moles of SiO2?
How many molecules are in 0.0125 moles of SiO2? 0.0 125 molso x 6.024 1023 molecules Jioz molecular sitamos sioz 13.
How many moles is 15.5 g SiO2?
About a 1/4 of a mole.
How much atoms are in a mole?
The value of the mole is equal to the number of atoms in exactly 12 grams of pure carbon-12. 12.00 g C-12 = 1 mol C-12 atoms = 6.022 × 1023 atoms • The number of particles in 1 mole is called Avogadro’s Number (6.0221421 x 1023).
What is Fe3O4 called?
Iron Oxide (fe3o4)



