What is the hybridisation of HNO2?
The Hybridization of HNO2 is Sp2.
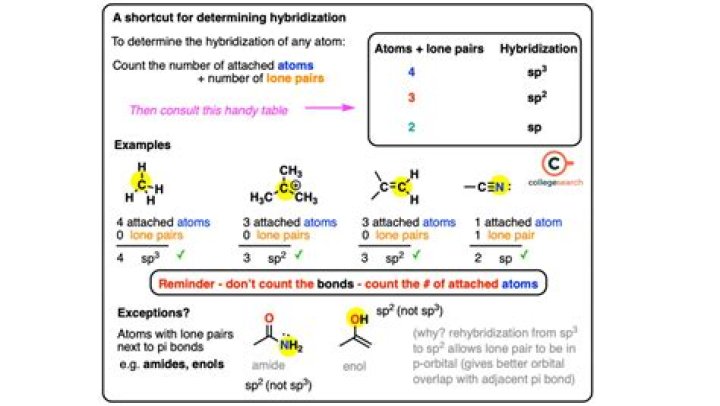
Hybridization is determined by adding the number of bonds and the lone pair of the central atom.
What is SP2 hybridization?
The sp2 hybridization is the mixing of one s and two p atomic orbitals, which involves the promotion of one electron in the s orbital to one of the 2p atomic orbitals. The new set of formed hybrid orbitals creates trigonal structures, creating a molecular geometry of 120 degrees.
Why is HNO3 trigonal planar?
The electron geometry is trigonal planar. The N atom is sp虏 hybridized. The O bonded to H has SN = 4 (two bonding pairs and two lone pairs). After bonding has occurred, some of the atomic orbitals have overlapped to form atomic orbitals.
What is the shape of NO2?
Except that NO2 is a V-shaped molecule, and CO2 is linear. Why are NO2 and CO2 different shapes? two N=O double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180掳 bond angle, and it is linear, as with CO2.
What is hybridization of N in NH3?
In Ammonia (NH3) or to be more precise the central atom in ammonia which is nitrogen is sp3 hybridized.
What is the hybridization of NH4+?
Q. In the ammonium ion NH4+, what percentage of p character do the hybrid orbitals of nitrogen possess? The nitrogen atom forms 4 bonds and has 0 lone pairs, so it needs 4 hybrid orbitals. Its hybridization state is therefore sp3.
What is sp3d2 hybridization explain with an example?
sp3d2 hybridization has 1s, 3p and 2d orbitals, that undergo intermixing to form 6 identical sp3d2 hybrid orbitals. These 6 orbitals are directed towards the corners of an octahedron. They are inclined at an angle of 90 degrees to one another.
What is sp3 hybridisation?
The term 鈥渟p3 hybridization鈥 refers to the mixing character of one 2s-orbital and three 2p-orbitals to create four hybrid orbitals with similar characteristics. In order for an atom to be sp3 hybridized, it must have an s orbital and three p orbitals.
Which has sp3 hybridization?
The bonds in a methane (CH4) molecule are formed by four separate but equivalent orbitals; a single 2s and three 2p orbitals of the carbon hybridize into four sp3 orbitals. In the ammonia molecule (NH3), 2s and 2p orbitals create four sp3hybrid orbitals, one of which is occupied by a lone pair of electrons.
Is HNO3 nonpolar or polar?
Nitric acid is polar in nature and it has a molecular geometry of nitric acid that is trigonal
How do you find the hybridization of NO2?
NO2 involves an sp2 type of hybridization. The most simple way to determine the hybridization of NO2 is by drawing the Lewis structure and counting the number of bonds and lone electron pairs around the nitrogen atom. You will find that in nitrogen dioxide there are 2 sigma bonds and 1 lone electron pair.
What shape is clo4?
The correct answer is Tetrahedral. Tetrahedral molecules array four atoms around a central atom, where every atom is oriented109.
Is NF3 tetrahedral?
The molecular geometry or shape of NF3 is a trigonal pyramid and its electron geometry is tetrahedral. NF3 lewis dot structure contains 1 lone pair and 3 bonded pairs.



