What is the electronic geometry of tef5 -?
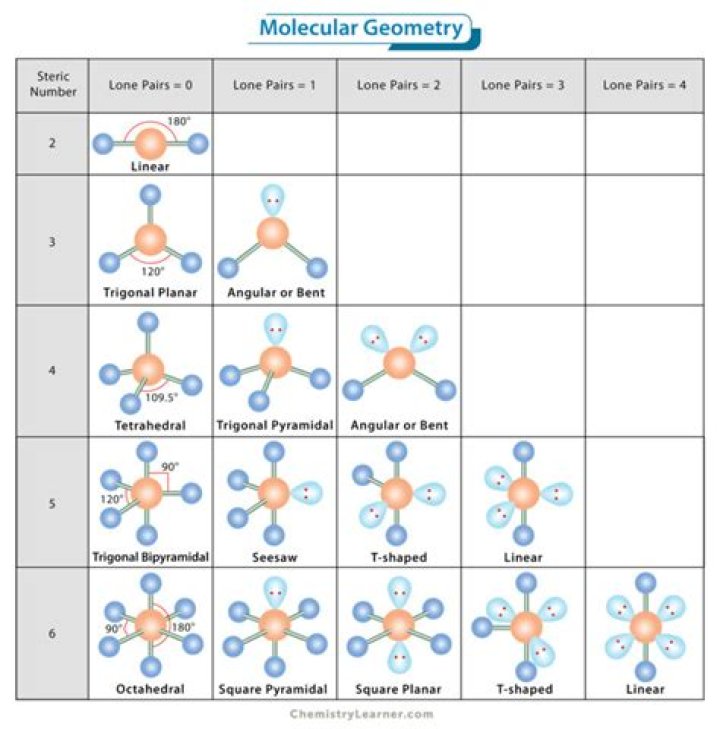
Electron domain geometry: octahedral.
Is tef5 polar or nonpolar?
This ion is considered to be polar.
How many right angles bond angles are present in tef5?
There are eight right angles in XeF+5 .
Is I3 linear?
I3- molecular geometry is linear. While there are three Iodine atoms, one of the atoms has a negative charge which further gives 3 lone pairs of electrons and 2 bond pairs. Its steric number will be 5. The three lone pairs will repel each other and take up equatorial positions.
Is AsF5 polar or nonpolar?
The molecular geometry of AsF5 is trigonal bipyramidal and its electron geometry is also the same. The lewis structure of AsF5 has 5 bonding pairs and 15 nonbonding pairs. AsF5 is a nonpolar molecule because of symmetrical geometry that makes the net dipole moment zero.
What are the bond angles in the following molecular model of pf5?
the pf5 bond angle will be 120 and 90 degree since it has a trigonal bipyramidal molecular geometry . As a result they will be pushed apart giving the pf5 molecule a trigonal bipyramid molecular geometry or shape. the pf5 bond angle will be 120 and 90 degree since it has a trigonal bipyramidal molecular geometry .
Which of the following has square pyramidal geometry?
Some molecular compounds that adopt square pyramidal geometry are XeOF4, and various halogen pentafluorides (XF5, where X = Cl, Br, I). Complexes of vanadium(IV), such as vanadyl acetylacetonate, [VO(acac)2], are square pyramidal (acac = acetylacetonate, the deprotonated anion of acetylacetone (2,4-pentanedione)).
What is the molecular geometry of XeF5?
Thus for [XeF5]- the parent shape is a pentagonal bipyramid with the lone pairs opposite to each other in order to minimise lone pair/lone pair repulsions. The [XeF5]- anion is therefore pentagonal planar. Check this for yourself by examining the crystal structure of this molecule (CSD refcode SOBWAH).
What kind of molecular geometry not electron group geometry !) Does boron trifluoride have?
The center boron atom of BF3 has no lone pairs of electrons, resulting in trigonal planar electron geometry. However, the molecular geometry of BF3 looks like a trigonal planar and no lone pairs on the top and bottom of the BF3 geometry. It’s the BF3 molecule’s symmetrical geometry.
Is I3 trigonal?
I3- has 2 bonds and 3 lone pairs for 5 regions of electron density. Placing that into a trigonal bipyramidal structure, the lone pairs go equitorial and the two other I atoms go axial. With the three atoms in a line, the molecule is linear.
Why is I3 polar?
I3- is formed by the reaction of I2 (iodine) with I- (iodide). When you add Iodine crystals to a solution of potassium iodide in water, you will generate a fair amount of KI3 (potassium triiodide). I3- is most definitely polar, since it is an anion and carries a negative charge.
Why is I3 not bent?
Since I3+ has sp3 hybridisation and two lone pair on central I atom therefore its shape would be Bent or V shape. Since I3+ has sp3 hybridisation and two lone pair on central I atom therefore its shape would be Bent or V shape. The tri-iodide ion has a total of 9 lone pairs.
What is the molecular geometry for the molecule IF5?
According to the VSEPR theory, the IF5 molecule ion possesses square pyramidal molecular geometry. Because the center atom, Iodine, has five I-F bonds with the five fluorine atoms surrounding it. The I-F bond angle is 82 degrees in the square pyramidal IF5 molecular geometry.
What is bonding group?
In the table below the term bonding groups/domains (second from the left column) is used in the column for the bonding pair of electrons. Groups is a more generic term. Group is used when a central atom has two terminal atoms bonded by single bonds and a terminal atom bonded with two pairs of electrons (a double bond).
How many double bonds are there in the hydrazine n2h4 molecule?
Now let’s talk about the N-N bond, each nitrogen has three single bonds and one lone pair. If we convert the lone pair into a covalent bond then nitrogen shared four bonds(two single and one double bond).



