What is the electron configuration of the V2+ ion?
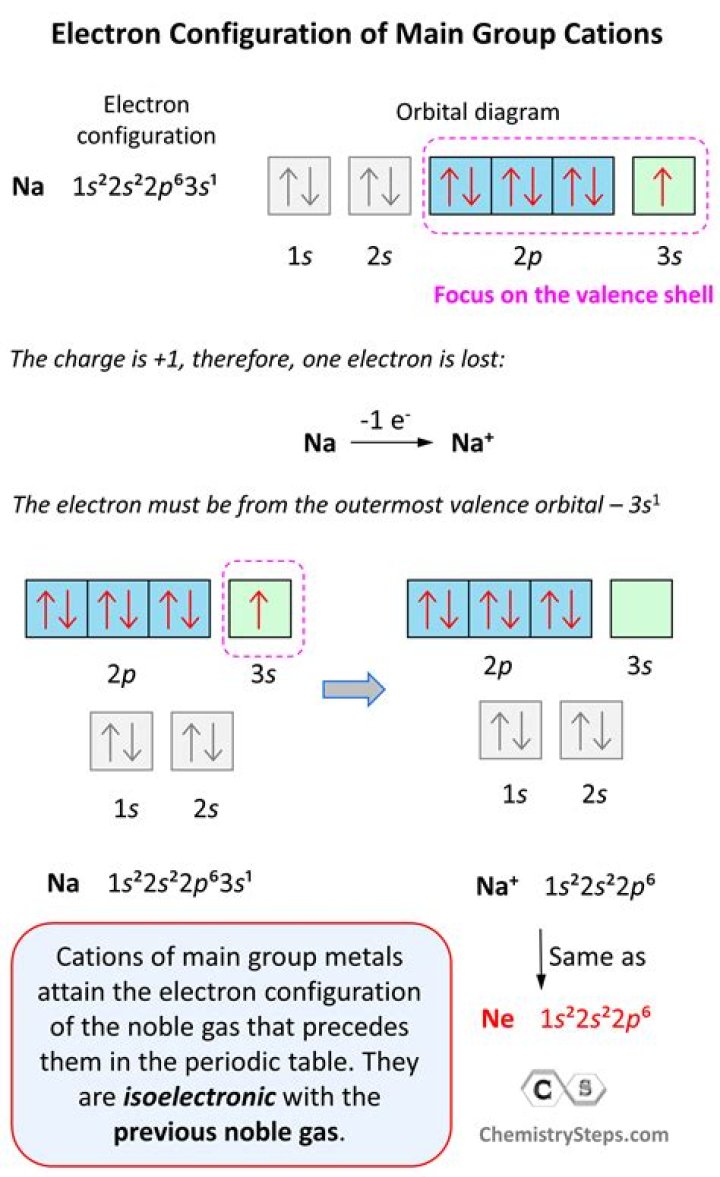
Configuration of V is [Ar]4s23d3. Hence electrons are removed from 4s. Thus configuration V2+ is [Ar]3d3.
What is the electron configuration of the V2+ ion?
1 Expert Answer
In shorthand, the electron configuration for V is [Ar]4s23d3 . However, when electrons are removed – I know it’s weird – the s-electrons come off first. So V2+ would be [Ar]3d3 .
How many electrons does V2+ have?
This electron configuration of vanadium ion(V2+) shows that vanadium ion has three shells and the last shell has eleven electrons. For this, the valence electrons of the vanadium ion(V2+) are eleven. On the other hand, The electron configuration of vanadium ions(V3+) is 1s2 2s2 2p6 3s2 3p6 3d2.
What is the ground-state electron configuration for V2 +?
VANADIUM (V)
V is atomic number 23 , so its configuration is 1s22s22p63s23p63d34s2 . In shorthand it is [Ar]3d34s2 .
What is the electron configuration of the V3+ ion?
In the V3+ ion, three electrons are removed and the configuration becomes [Ar]3d24s0 [ A r ] 3 d 2 4 s 0 . Hence the condensed ground-state electronic configuration of V3+ ion is [Ar]3d2 [ A r ] 3 d 2 .
What is the electronic configuration of vanadium Z 23?
Atomic number of V = 23 and thus the electronic configuration = [Ar]3d34s2.
What is the electron configuration of iodine I )?
The ground state electronic configuration of Neutral Iodine atom is [Kr] 4d10 5s2 5p5.
Why the electronic configuration of potassium is 2881?
s can hold 2 electrons,p can hold 6 electrons,d can hold 10 electrons,f can hold 14 electrons, and so on. Now, K= 1s2 2s2 2p6 3s2 3p6 4s1. So, we see that 3d has more energy than 4s. Hence, it enters the 4th shell, for the configuration of 2,8,8,1.
What is the electronic configuration of potassium class 9?
The electronic configuration of potassium is 2,8,8,1 and not 2,8,9.



