What is the difference between interdiffusion and self-diffusion?
Self-diffusion is atomic migration in pure metals (all atoms exchanging positions are of the same type). Interdiffusion is diffusion of atoms of one metal into another metal.
What is the difference between interdiffusion and self-diffusion?
Interdiffusion is diffusion of atoms of one metal into another metal. Self-diffusion involves the motion of atoms that are all of the same type, therefore it is not subject to observation by composition changes, as with interdiffusion.
What is self-diffusion interdiffusion?
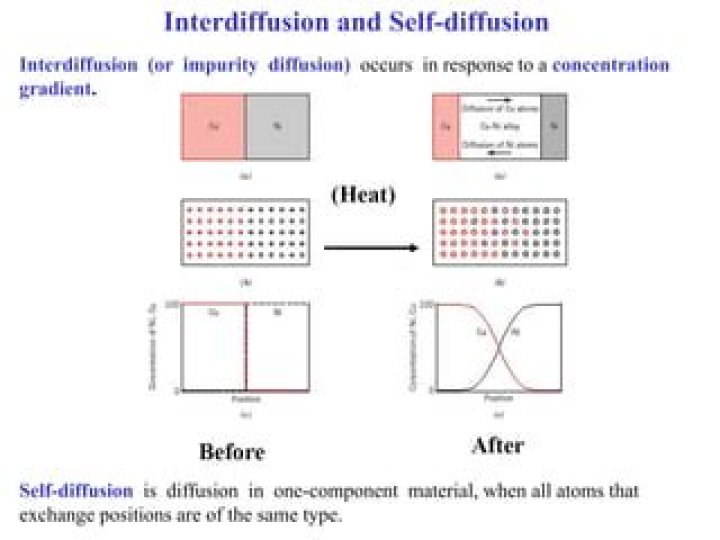
Interdiffusion (or impurity diffusion) occurs in response to a concentration. gradient. Self-diffusion is diffusion in one-component material, when all atoms that. exchange positions are of the same type.
What is meant by self-diffusion?
The migration of constituent atoms or molecules within the bulk of a substance, especially in a crystalline solid.
What are the differences between interstitial and vacancy diffusion?
The unit step in vacancy diffusion is an atom breaks its bonds and jumps into neighboring vacant site. In interstitial diffusion, solute atoms which are small enough to occupy interstitial sites diffuse by jumping from one interstitial site to another.
How do you monitor self-diffusion?
Self-diffusion may be monitored by using radioactive isotopes of the metal being studied. The motion of these isotopic atoms may be monitored by measurement of radioactivity level.
What is vacancy diffusion?
Vacancy diffusion (Fig. 1) is the predominant diffusion mechanism in metals due to the low energy required to move atoms into atomic vacancies that form during heating. The vacancy diffusion process occurs when an atom on a normal lattice site jumps into an adjacent unoccupied (vacant) site.
What is the interdiffusion coefficient?
The interdiffusion coefficient is usually a composition-dependent quantity. On the other hand, interdiffusion is due to the diffusive motion of A and B atoms, which in general have different intrinsic diffusion coef- ficients.
What is diffusion in solids liquids and gases?
Spreading out and Mixing of one substance with other is called Diffusion. It happens in all forms of matter – Solids, Liquids and Gases. It is fastest in gases and slowest in solids.
Why is diffusion in solids important?
Diffusion is an important mode of mass transport in solids where atoms move to form compounds and find lattice positions. Charges and defects can also diffuse. random motion and concentration dependence are introduced via simple derivations of Fick’s laws for diffusion.
What is self-diffusion in physics?
Self-diffusion is displacement of molecules due to Brownian motion in a medium of identical molecules. Tracer diffusion is the same phenomena defined for multi-component systems and is the case when displacement of a molecule in a mixture is monitored.
What is Fick’s second law of diffusion?
Fick’s 2nd law of diffusion describes the rate of accumulation (or depletion) of concentration within the volume as proportional to the local curvature of the concentration gradient.
What is binary diffusion?
binary fission, asexual reproduction by a separation of the body into two new bodies. In the process of binary fission, an organism duplicates its genetic material, or deoxyribonucleic acid (DNA), and then divides into two parts (cytokinesis), with each new organism receiving one copy of DNA.
What is diffusion and explain diffusion mechanisms in detail?
Diffusion is the process of mixing which involves the movement of atoms from area of higher to those of lower concentration. or. Diffusion is the shifting of atoms and molecules to new sites within a material resulting in the uniformity of composition as a result of thermal agitation.
What is diffusion and explain diffusion mechanisms in detail write the names of the factors that influence diffusion?
What are the factors affecting diffusion? Temperature, area of interaction, size of the particle and the steepness of the concentration gradient are all factors that affect the process of diffusion.
What is steady and non steady state diffusion process?
Steady State Diffusion: Steady state diffusion is a form of diffusion that takes place at a constant rate. Unsteady State Diffusion: Unsteady state diffusion or non-steady state diffusion is a form of diffusion in which the rate of diffusion is a function of time.
What is diffusion profile?
After annealing for a specified time at a specified temperatures, some diffusion of the impurity atoms will have produced a diffusion profile, i.e. a smooth curve of the concentration c vs. depth x in the sample (usually plotted as lg(c) – x curve).
What are the different diffusion profiles obtained in the diffusion?
8.2 Diffusion Profiles
The diffusion profile of dopant atoms is dependent on the initial and boundary conditions. Solutions for Equation 8.3 have been obtained for various simple conditions, including constant-surface-concentration diffusion and constant-total- dopant diffusion.
Which of the following is not true for diffusion flux *?
The correct answer is (c) Rate of diffusion of gases depends on their volume. Rate of diffusion of gases does not depends on their volume.



