What is the coordination number of fcc and CCP?
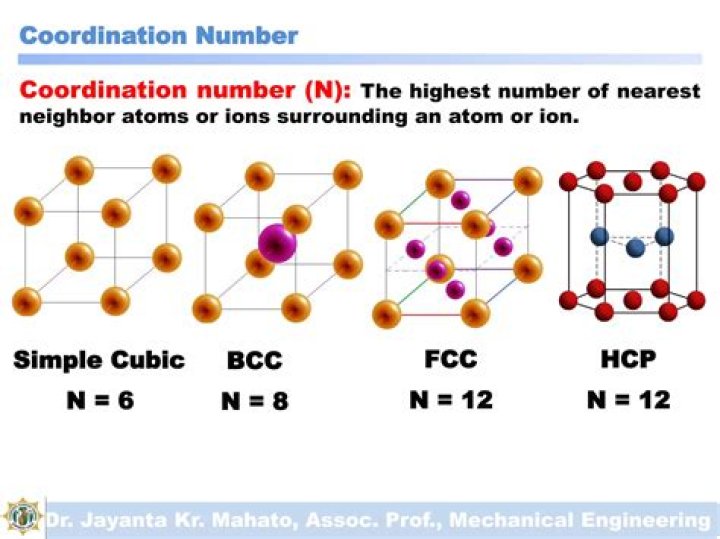
Coordination number – the number of nearest neighbor atoms or ions surrounding an atom or ion. For FCC and HCP systems, the coordination number is 12.
What is the coordination number of fcc and CCP?
-fcc stands for face centered cell and the coordination number is 12. -ccp stands for cubic close packed and the coordination number is 12.
How does fcc have a coordination number of 12?
Face Centered Cubic (FCC) Structure
Each of the corner atoms is the corner of another cube so the corner atoms are shared among eight unit cells. Additionally, each of its six face centered atoms is shared with an adjacent atom. Since 12 of its atoms are shared, it is said to have a coordination number of 12.
How do you find the coordination number?
Here are the steps for identifying the coordination number of a coordination compound.
Identify the central atom in the chemical formula. Locate the atom, molecule, or ion nearest the central metal atom. Add the number of atoms of the nearest atom/molecule/ions. Find the total number of nearest atoms.
What is coordination number of atoms in CCP and bcc?
The co-ordination number of atoms in BCC lattice is 8, in HCP lattice 12, in CCP lattice 12 and in simple lattice it is 6.
What is meant by coordination number?
coordination number, also called Ligancy, the number of atoms, ions, or molecules that a central atom or ion holds as its nearest neighbours in a complex or coordination compound or in a crystal.
Why is the coordination number of HCP 12?
HCP means hexagonal close packed. The repeating unit in a crystal structure is called a unit cell. Complete step by step answer: – Therefore there are 12 atoms in touch or coordination with each atom in a Hexagonal close packed structure, then the coordination number of HCP is 12.
What is 2nd coordination number?
The second coordination number of an atom is the number of atoms that are second closest to it. In a primitive cubic unit cell, the closest atoms are at a distance of 1, i.e. the edge length.
Why is the coordination number of simple cubic 6?
This structure is called simple cubic packing. Each sphere in this structure touches four identical spheres in the same plane. Each atom in this structure can form bonds to its six nearest neighbors. Each sphere is therefore said to have a coordination number of 6.
What is the coordination number of CR in K3 Cr Ox 3?
Coordination number of Cr is six.
What is coordination number in solid state?
Co-ordination number is the number of spheres (atoms, molecules or ions) directly surrounding a single sphere in a crystal. The coordination number in a face centered cubic close packing structure is 12.
What is coordination number Write the coordination number of HCP and CCP?
A CCP arrangement has a total of 4 spheres per unit cell and an HCP arrangement has 8 spheres per unit cell. However, both configurations have a coordination number of 12.
Is fcc the same as CCP?
Face Centered Cubic (fcc) or Cubic Close Packed (ccp) These are two different names for the same lattice. We can think of this cell as being made by inserting another atom into each face of the simple cubic lattice – hence the “face centered cubic” name.
How do you find the bcc coordination number?
In the bcc structure each atom has c1=8 c 1 = 8 nearest neighbours (coordination number) at a distance of dc1=2r=√32a≈0.866a(3) (3) d c 1 = 2 r = 3 2 a ≈ 0.866 a and c2=6 c 2 = 6 next-nearest neighbours at a distance of dc2=a≈2.3r≈1.15dc1.
What is the coordination number of chromium in K3 Cr C2O4 3?
The IUPAC name of K3[Cr(C2O4)3] = Potassium trioxalato chromate (III). The coordination number will be 6 as it is bonded to three bi-dentate ligands.
What is coordination number give two examples of complexes having coordination number of 2 and 4?
Some examples include: [Ag(NH3)2]+ where Ag has coordination number 2 and the molecular geometry of the compound is linear. [NiCl4]2− where Ni has coordination number 4 and the molecular geometry of the compound is square planar.
What is the coordination number of Fe in fecl2 en 2 Cl?
Coordination number of Fe and Co in [Fe(C2O4)3]3− and [Co(en)3]3+ respectively is 6.



