what is the charge of the nucleus of element 89, check these out | What is the charge of the nucleus of an element?
The number of protons corresponds to the charge of the nucleus since neutrons have no net charge. The charge of the nucleus is 89+
What is the charge of the nucleus of an element?
A nucleus of an atom has protons and neutrons. We know that a proton has a charge of +1 , while a neutron has no charge, or 0 . Therefore, the nucleus of an atom will always have a positive charge.
What is the charge of the nucleus of element 44?
Explanation: Ruthenium metal has Z=44 . What does this mean? It means that element we call ruthenium has 44 massive, formally positively charged particles in its nucleus, 44 protons , and that number defines the nucleus as ruthenium.
What is the charge of the nucleus of element 92?
The nucleus, therefore, has a combined mass of 238 nucleons, and a charge of +92. The beginning of the map shows how protons and neutrons agglomerate in order to form light nuclei, hydrogen, helium, lithium and berylium isotopes.
What is nucleus in chemistry class 9?
Hint: The nucleus is the solid mass present in the center of the atom which consists of two types of particles , out of which one type of particle carries the positive charge and the other type of particles carries the no charge.
What is the charge of the nucleus of element 89 quizlet?
What is the charge of the nucleus of Element 89? Because it is the nucleus only this element has a charge of positive 89.
What element has the atomic number 76?
Osmium – Element information, properties and uses | Periodic Table.
What element has an atomic number of 56?
Barium – Element information, properties and uses | Periodic Table.
What is the use of uranium-235?
What is it used for? Uranium “enriched” into U-235 concentrations can be used as fuel for nuclear power plants and the nuclear reactors that run naval ships and submarines. It also can be used in nuclear weapons.
What is the total nuclear mass of U-235?
2. Uranium-235 has a nuclear mass of 235.0439299 u. It contains 92 protons.
What is the protons of uranium 238?
Uranium has an atomic number of 92 which means there are 92 protons and 92 electrons in the atomic structure. U-238 has 146 neutrons in the nucleus, but the number of neutrons can vary from 141 to 146. Because uranium is radioactive, it is constantly emitting particles and changing into other elements.
What is nucleus in chemistry Byjus?
The nucleus of an atom is the central region of an atom where the majority of the mass is concentrated. Through the scattering of alpha particles experiment by Rutherford, we learned that the nucleus of an atom contains a majority of the mass of the atom.
What is nucleus in chemistry for Class 6?
The nucleus is the command centre of a cell. This is because it contains the genetic material of the cell. Therefore, it consists of a number of structural elements which facilitate its functions. The nucleus of a cell has a spherical shape.
What is nucleus of an atom Class 10?
The nucleus, that dense central core of the atom, contains both protons and neutrons. Electrons are outside the nucleus in energy levels. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge.
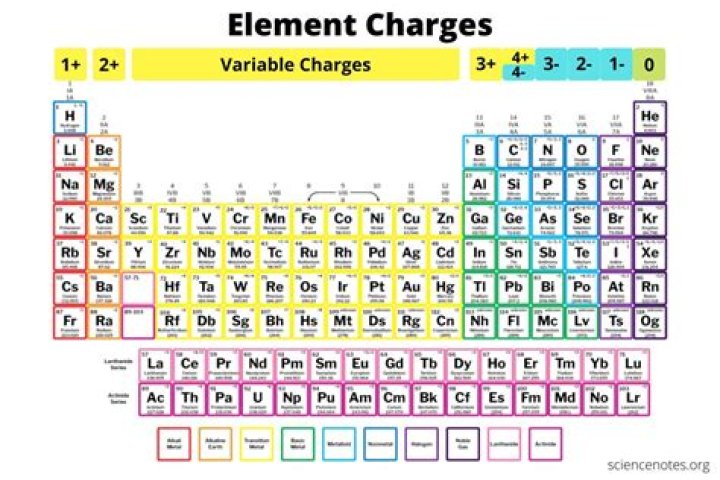
How do you figure out the charge of an element?
For a single atom, the charge is the number of protons minus the number of electrons.



