What is the bond angle for the central atom in SeO3?
Hence, the polyatomic molecule SeO3 S e O 3 has a trigonal planar geometry with a 120 degree bond angle.
What is the bond angle for the central atom in SeO3?
Sulphur trioxide is also non-polar. It has a bond angle of 120o.
What is the molecular geometry of CH3?
CH3- is trigonal pyramidal with bond angles of slightly less than 109.5 degrees because of the lone pair, like the other molecules you mentioned.
What is the electron geometry of if4 +?
The molecular geometry of the ion, which does not take into account the lone pairs present on the central atom, will be square planar, AX4E2 . The electron-domain arrangement, which does include any lone pairs attached to the central atom, will be octahedral.
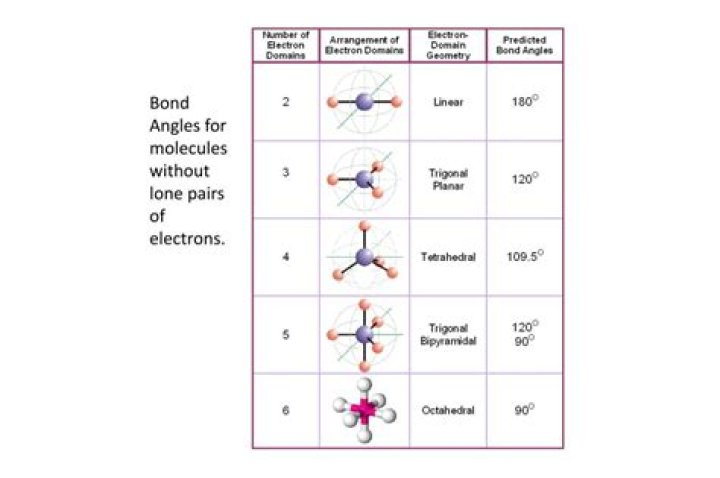
What are the angles in a trigonal planar geometry?
Trigonal planar: triangular and in one plane, with bond angles of 120°. Tetrahedral: four bonds on one central atom with bond angles of 109.5°.
How many lone pairs are drawn in the best Lewis structure for SeO3 2?
By In both Lewis structures, there are eight lone pairs on all oxygen atoms.
Is SeO2 a double covalent bond?
The selenium dioxide molecule has a central Se atom to which two oxygen atoms are bonded. The bonds are double bonds for which four of its valence
What is the shape and bond angle of CH3?
The carbon atom in the ion CH+3 is sp2 hybridized carbon and therefore, the geometry around this atom is trigonal planar. In this case, the H−C−H bond angle is 120∘ .
Does CH3 have tetrahedral bond angles?
Each CH 3 group is AX4; its electron-group arrangement is tetrahedral. The third C is AX3, a trigonal planar arrangement. The CH3 groups should have about 109.5° bond angles.
What is the molecular geometry of CH4?
For example; four electron pairs are distributed in a tetrahedral shape. If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4).



