What is pf5 molecular geometry?
The molecular geometry of PF5 is trigonal bipyramidal with symmetric charge distribution. Therefore this molecule is nonpolar. Phosphorus Pentafluoride on Wikipedia.
What is the electron geometry and molecular shape of PF5?
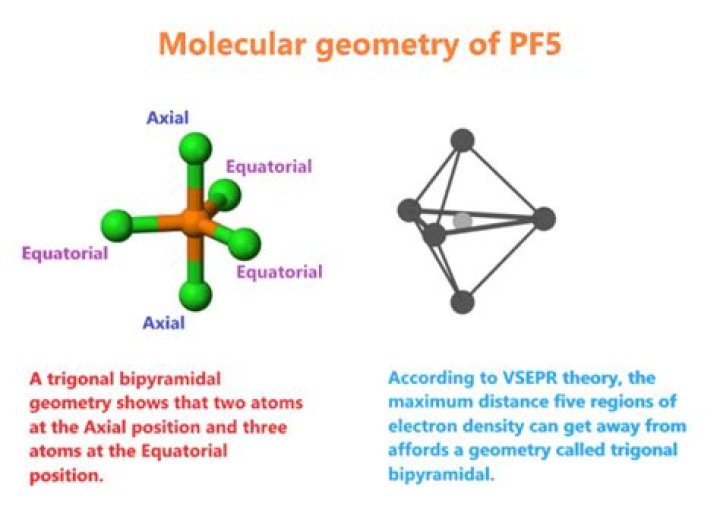
Phosphorus pentafluoride has 5 regions of electron density around the central phosphorus atom (5 bonds, no lone pairs). The resulting shape is a trigonal bipyramidal in which three fluorine atoms occupy equatorial and two occupy axial positions.
Which is the best description of the molecular geometry of the molecule PF5?
In PF5 there is a lone pair of electrons on phosphorus where as in IF5 there are no lone pairs on iodine. As a result, the molecular shape of PF5 is square pyramidal and IF5 is trigonal bipyramidal.
PF5 Molecular Geometry
Phosphorus forms single bonds with all five Fluorine atoms, which means there are five regions of electrons density. This arrangement of the atoms makes the molecular geometry of PF5 Trigonal Bipyramidal.
What is PF5 is a polar molecule?
Is PF5 a polar or nonpolar molecule? PF5 is a nonpolar molecule because there is no net dipole moment in the molecule. As the shape of the molecule is symmetric with even charge distribution, it makes Phosphorus Pentafluoride a nonpolar molecule.
A) The shape of PCl5 is trigonal bipyramidal but of ClF3 is ‘T’ shaped, although in both molecules central atom has sp3. d hybridization, explain.
Does PCl5 have regular geometry?
here p atom undergo sp3d hybridisation and form 5 equivalent sp3d orbitals, each contaisn an unpair electrons . the geometry of its is also regular and its trigonal pyramidal structure by overlapping of orbitals .
What will be the geometry and hybridization of PCl5?
Hybridization : Geometry of PCl5 molecule is trigonal bipyramidal.
Phosphorus has 5 valence electrons. 1 electron each is shared by chlorine atom hence, PCl5 molecule has sp3d hybridisation and hence trigonal bipyramidal shape.
What is the bond polarity of PCl5?
PCl5 is nonpolar in nature because it has the symmetrical geometrical structure due to which the polarity of P-Cl bonds gets canceled by each other. As a result, the net dipole moment of PCl5 comes out to be zero.
How many atoms does PF5?
PF5 crystallizes in the hexagonal P6_3/mmc space group. The structure is zero-dimensional and consists of two phosphorus pentafluoride molecules. P5+ is bonded in a trigonal bipyramidal geometry to five F1- atoms. There is three shorter (1.56 Å) and two longer (1.61 Å) P–F bond length.
What is the orbital hybridization in PF5?
The hybridization is sp3d hybridization and phosphorous atom forms five sp3d hybrid orbitals. Five hybrid orbitals will be used to form bonds with five fluorine atoms.
So a total of sp3d5 hybridized orbitals are formed. The five sp3d hybrid orbitals of P bond with five F atoms forming five O-F sigma bonds. So in the molecule, there are five bond pairs and no lone pairs and the molecule type is AB5. Note:Since the molecule type is AB5, it will have trigonal bipyramidal geometry.
Is PF5 ionic or molecular?
Thus, PF5 has net four covalent bonds and one ionic bond.
How do the molecular geometry of the molecule IF5 and PF5 differ?
How do the molecular shapes differ? In IF5 there is a lone pair of electrons on iodine where as in a PF5 there are no lone pairs on phosphorus. As a result, the molecular shape of IF5 is square pyramidal and PF5 is trigonal bipyramidal.



