What is geometry of BrF5?
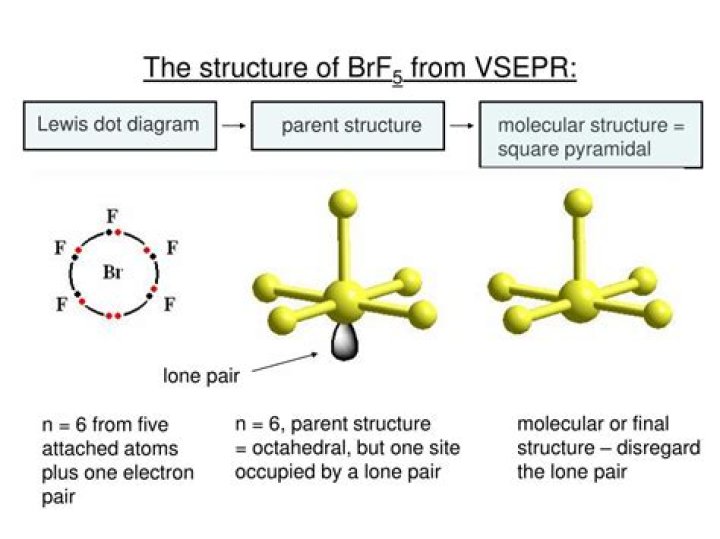
The molecular geometry of BrF5 is square pyramidal and its electron geometry is octahedral.
Is brf3 sp3 hybridized?
The hybridization that takes place in BrF3 is sp3d. We will understand how hybridization of BrF3 occurs in the molecules as well as its molecular geometry and the bond angles below.
In which of the following central atom is sp3d hybridised BrF5?
In BrF5 molecule, bromine atom has sp3d2 hybridisation. It is square pyrmidal in shape due to the presence of one pair of electrons.
Is BrF5 square pyramid?
The molecules like BrF5 and XeOF4 are square pyramidal in shape.
What is the hybridization of bf3?
BF3 is an sp2 hybridization. It is sp2 for this molecule because one 蟺 (pi) bond is needed for the double bond between the Boron, and just three 蟽 bonds are produced per Boron atom. The atomic S and P 鈥 orbitals in Boron outer shell mix to form three equivalent hybrid orbitals of sp2.
Is BrF3 ionic or covalent?
When the difference in the electronegativity between two elements is from 0 to 0.4 then the bond is nonpolar covalent. Here, we have to calculate the difference in the electronegativity of BrF3 B r F 3 molecule. Therefore, the bonds of BrF3 B r F 3 molecule are Polar Covalent.
Why BrF5 is sp3d2?
BrF5: Br is attached to 5 F atoms through 5 sigma bonds and there is a lone electron pair. So, steric number is 6 and hence, Br atom is sp3d2 hybridized in BrF5. The electron pair geometry is octahedral, but molecular shape is square pyramidal.
How many electron groups are in BrF5?
Hence, there are 6 electron groups present around the central atom in bromine pentafluoride.
What is the hybridization of bro4?
The hybridization of Br in BrO鈭4 B r O 4 鈭 is sp2 s p 2 and it has tetrahedral structure.
Which has sp2 hybridization?
Sulphur in SO2, is sp2-hybridised.
What happens when BrF5 is hydrolyzed?
Bromine pentafluoride reacts with water to form bromic acid and hydrofluoric acid (especially when moderated by dilution with acetonitrile): BrF5 + 3 H2O 鈫 HBrO3 + 5 HF.
What is hybridization sp2 sp3 hybridisation?
sp hybridization occurs due to the mixing of one s and one p atomic orbital, sp2 hybridization is the mixing of one s and two p atomic orbitals and sp3 hybridization is the mixing of one s and three p atomic orbitals.
What is the molecular geometry around the carbon in CH3OH?
These two lone pairs on the O atom cause repulsion within the CH3OH molecule. Thus instead of tetrahedral, CH3OH forms also forms a bent shape. Around the Carbon atom, it shows a tetrahedral geometry with 3 hydrogen bonds and 1 hydroxyl bond.



