What is bleach on the pH scale?
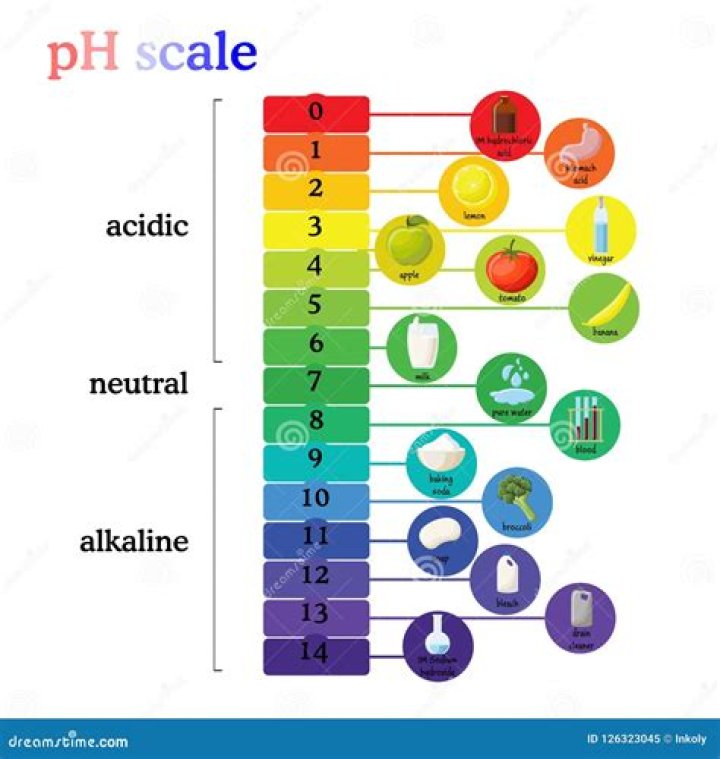
Bleach: pH 11-13. Bleach is one of the most common cleaning supplies in households and commercial settings. This particular product has a pH between 11 and 13. Its high level of alkalinity is what makes it corrosive.
Is bleach a base or acid?
Chlorine bleach is a base and is especially good at removing stains and dyes from clothes as well as disinfecting.
Is bleach an acid or alkaline or neutral?
For domestic use, bleach usually contains 5% sodium hypochlorite, giving it a pH of around 11 and making it mildly irritating to the skin. Concentrated bleach (10-15% sodium hypochlorite) is highly alkaline (pH ~13) and now is now so corrosive that it can burn skin on contact.
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Hypochlorous acid (HOCl or HClO) is a weak acid that forms when chlorine dissolves in water, and itself partially dissociates, forming hypochlorite, ClO−.
Is chlorine acid or alkaline?
When chlorine (in any form) is added to water, a weak acid called Hypochlorous acid is produced. It is this acid, not the chlorine, which gives water its ability to oxidize and disinfect. Proper chlorination and filtration give pool water its clear, sparkling appearance. Chlorine exists as a solid, a liquid and a gas.
Bleach is one of the most common cleaning supplies in households and commercial settings. This particular product has a pH between 11 and 13. Its high level of alkalinity is what makes it corrosive.
What pH scale is vinegar?
Vinegar is acidic. Vinegar’s pH level varies based upon the type of vinegar it is. White distilled vinegar, the kind best suited for household cleaning, typically has a pH of around 2.5. Vinegar, which means “sour wine” in French, can be made from anything containing sugar, such as fruit.
Does bleach change pH?
Bleach is basically ph neutral. It will raise the ph slightly when added but as the chlorine get used up, it will drop back down. Some of the cheaper stuff, and the higher concentration stuff have more lye, which will raise ph a littlebut again, in most cases, ph neutral (long term).
When added to water, liquid chlorine (which has a pH of 13) makes HOCl (hypochlorous acid – the killing form of chlorine) and NaOH (sodium hydroxide), which raises pH.
What is the pH of lemon?
Lemon juice contains citric acid and has a pH of around 3.
What pH is soap?
Results: Majority of the soaps have a pH within the range of 9-10. Majority of the shampoos have a pH within the range of 6-7. Conclusions: The soaps and shampoos commonly used by the population at large have a pH outside the range of normal skin and hair pH values.
What color does pH paper turn with bleach?
The main ingredient in household bleach is sodium hypochlorite. This is usually diluted with water to a concentration of approximately 5 percent. The pH of this solution is approximately 11. This means that it is very alkaline and pH paper will turn dark blue/purple when it makes contact with household bleach.
The abbreviation pH stands for potential hydrogen, and it tells us how much hydrogen is in liquids—and how active the hydrogen ion is.
What is the pH of hypochlorous acid?
hypochlorous acid is presented from pH 5.0 to pH 8.0. Sodium hypochlorite (NaOCl) of the highest purity was prepared for this work.
What is the pH of sodium hypochlorite?
Sodium hypochlorite (NaOCl) in the form of laundry bleach is available in most households. The concentrate is about 5.25 to 6 percent NaOCl, and the pH value is about 12. Sodium hypochlorite is stable for many months at this high alkaline pH value.
How do you make sodium hypochlorite at home?
Sodium hypochlorite can be produced in two ways: – By dissolving salt in softened water, which results in a concentrated brine solution. The solution is electrolyzed and forms a sodium hypochlorite solution in water. This solution contains 150 g active chlorine (Cl2) per liter.



