What happens to the kinetic energy of the molecules?
The kinetic molecular theory can be used. Temperature is increased, so the average kinetic energy and the rms speed should also increase. This means that the gas molecules will hit the container walls more frequently and with greater force because they are all moving faster. This should increase the pressure.
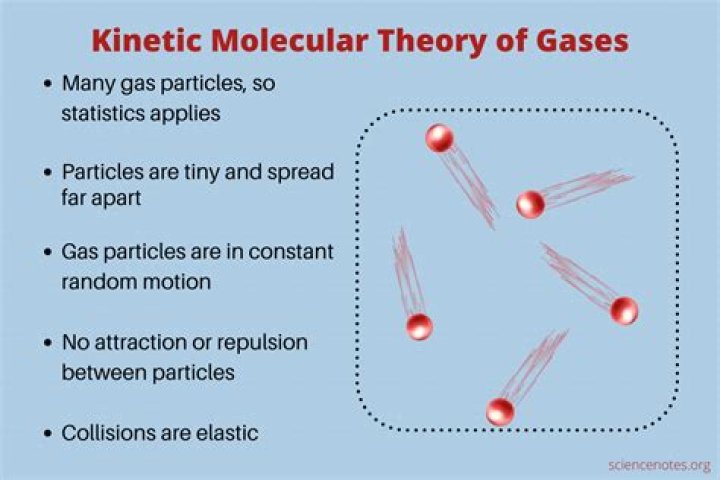
Which of the following statements about the kinetic molecular theory is true?
According to the kinetic molecular theory of gases, the size of the particle is significantly small when compared to the volume of the container. Since they’re in constant motion, the distance between molecules is much larger than the diameter of each molecule. The statement is true.
What is the kinetic energy of molecules?
The average translational kinetic energy of a molecule is called thermal energy.
What does kinetic energy of molecules depend on?
1). As such, it can be concluded that the average kinetic energy of the molecules in a thermalized sample of gas depends only on the temperature. However, the average speed depends on the molecular mass. So, for a given temperature, light molecules will travel faster on average than heavier molecules.
How does the kinetic energy of a molecule in a substance in with the decrease in temperature?
When a substance is cooled, the velocity of its molecules decreases. As a result of this, the kinetic energy of the molecule also decreases.
When the kinetic energy of molecules increases the molecules are moving?
In the kinetic theory of gasses, increasing the temperature of a gas increases in average kinetic energy of the molecules, causing increased motion. This increased motion increases the outward pressure of the gas, an expected result from the ideal gas equation PV=NkT.
Which one of the following statements is not true about the kinetic molecular theory of gases?
Answer: The following statement “The average kinetic energies of different gases are different at the same temperature.” is not consistent with the kinetic-molecular theory of gases.
Which statement is true about the kinetic molecular theory of gases?
The answer is b) The collisions of particles with one another are completely elastic.
Which statement is true about the average kinetic energy of a gas and its temperature?
At a given temperature, the average kinetic energy is proportional to that temperature. This means that large molecules have the same average kinetic energy as small molecules under the equipartition theorem.
What is kinetic energy describe kinetic molecular theory?
Kinetic energy is energy that an object has because of its motion. The Kinetic Molecular Theory explains the forces between molecules and the energy that they possess. When the molecules collide with each other, or with the walls of a container, there is no significant loss of energy.
How do you find mean kinetic energy?
The mean temperature of an ideal gas is related to the kinetic energy via E=3/2* k*T, where k is the Boltzmann constant (1.38*10^-23 J/K) and T is the Kelvin temperature equal to 273.15+ Celsius temperature.
What does the kinetic theory state about the kinetic energy of gas molecules?
Kinetic Molecular Theory states that gas particles are in constant motion and exhibit perfectly elastic collisions. The average kinetic energy of a collection of gas particles is directly proportional to absolute temperature only.
What happens to the kinetic energy and what happens to the momentum of the molecule?
What happens to the kinetic energy and what happens to the momentum of the molecule? Kinetic energy changes; momentum stays the same.
Which is true for internal energy?
Which one is true for internal energy? Explanation: All are correct for internal energy and are part of its property. Explanation: Internal energy does not depend on path.
Which phase has the greatest kinetic energy?
Energy and State of Matter
A pure substance in the gaseous state contains more energy than in the liquid state, which in turn contains more energy than in the solid state. Particles has the highest kinetic energy when they are in the gaseous state.
How does the kinetic energy of a molecule in a substance changes with increase in temperature?
Answer: increasing temperature always creates randomness in molecules of matterso increasing randomness promotes velocity of particles.and than we know about that kinetic energy is directly proportional to square of magnitude of velocity ( not velocity because velocity is a vector quantity).
How does the kinetic energy of the atoms of a substance affect its temperature?
The atoms and molecules in H have a higher average KE than those in L. The temperature of substance H will decrease, as will the average KE of its molecules; the temperature of substance L will increase, along with the average KE of its molecules.



