What elements have a positive charge?
Positive ions are cations and are typically metals like copper or sodium. Negatively-charged ions are anions, formed from nonmetallic elements like oxygen and sulfur.
What types of elements form positive ions?
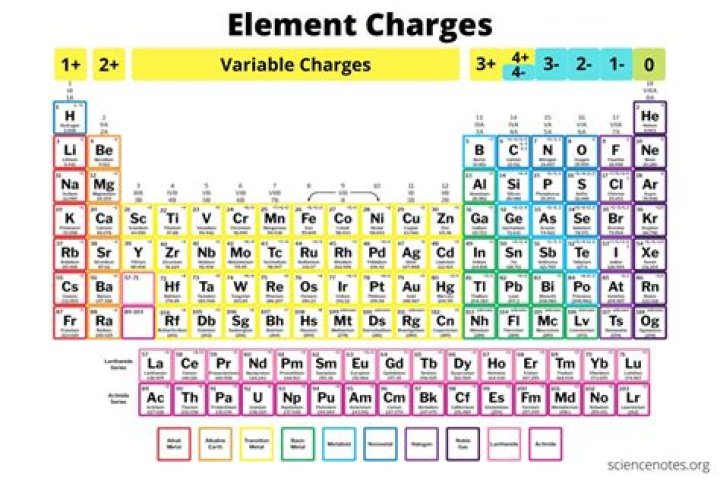
Metal elements form positively charged ions called cations because they are located on the left side of the periodic table. Groups 1 and 2 are called the alkali metals and alkaline Earth metals, respectively. These elements all have valence electrons in an s orbital.
How do you know which elements are positive and negative?
To find the ionic charge of an element you’ll need to consult your Periodic Table. On the Periodic Table metals (found on the left of the table) will be positive. Non-metals (found on the right) will be negative.
A cation is a positively charged ion with fewer electrons than protons while an anion is negatively charged with more electrons than protons. Because of their opposite electric charges, cations and anions attract each other and readily form ionic compounds.
What is positively charged particle?
A proton is a positively charged particle located in the nucleus of an atom. If it gains an extra electron, it becomes negatively charged and is known as an anion. If it loses an electron, it becomes positively charged and is known as a cation.
A cation has more protons than electrons, consequently giving it a net positive charge.
What is an example of a positive ion?
Examples of Positive Ions
Positive ions are typically metals or act like metals. Many common materials contain these ions. Mercury is found in thermometers, for instance, and aluminum is a metal that is found in a surprising amount of things. It’s even an ingredient in baking soda and in certain other food products!
What is a positive charge?
A positive charge occurs when the number of protons exceeds the number of electrons. A positive charge may be created by adding protons to an atom or object with a neutral charge.
Positively charged particles that are contained in the nucleus of the atom (the centre) they have a mass of 1amu (atomic mass unit).
Which of the following has a +ve charge?
Answer: The particles that exist inside the nucleus are protons and neutrons but only proton has the positive charge equal to 1.
Which element would be a positive ion in a compound?
Positive ions are cations and are typically metals like copper or sodium. Negatively-charged ions are anions, formed from nonmetallic elements like oxygen and sulfur.
What are positive and negative ions?
Ions are invisible charged particles in the air – either molecules or atoms, which bear an electric charge. To put it simply, positive ions are molecules that have lost one or more electrons whereas negative ions are actually oxygen atoms with extra-negatively-charged electrons.
There are three subatomic particles: protons, neutrons and electrons. Two of the subatomic particles have electrical charges: protons have a positive charge while electrons have a negative charge.



