What are the quantum numbers for the valence electrons in aluminum?
The valence electrons all have principle quantum number 3, so you tell us the number…….. As a consequence most of the chemistry of aluminum deals with Al3+ ……….
What is the principal quantum number of aluminium?
In your case, the principal quantum number, n , which gives the energy level on which the electron is located, is equal to 3 . Because in the case of an aluminium atom the p subshell contains a single electron, you can pretty much pick any of these three values for ml .
What are the valence electrons of aluminium?
Aluminium has 3 valence electrons. Valence electrons are located in the outermost shell of an element.
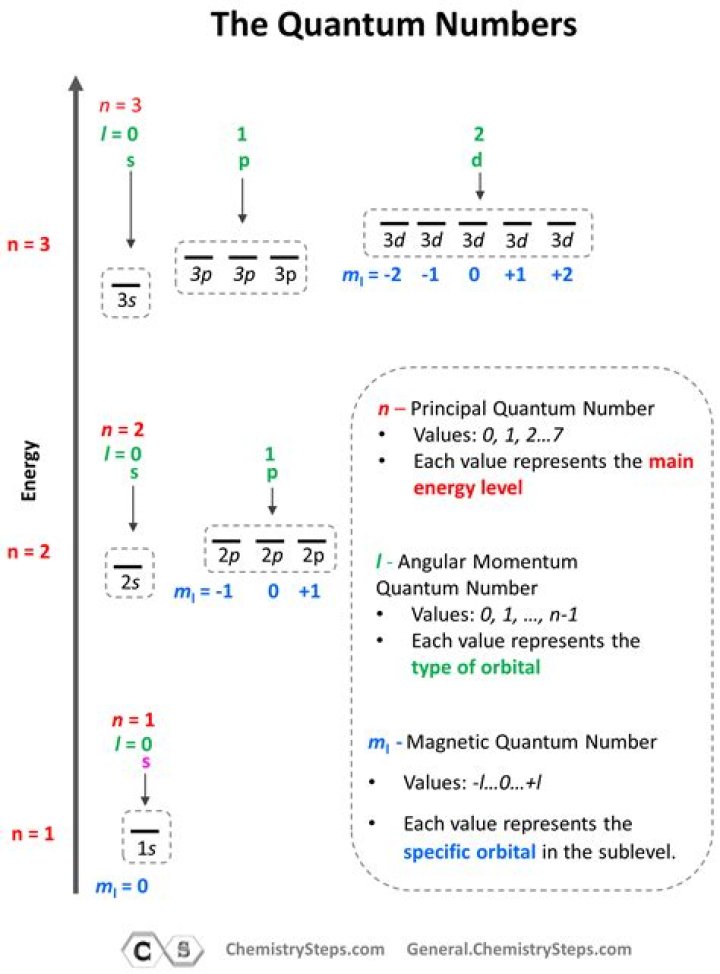
Quantum Numbers
To completely describe an electron in an atom, four quantum numbers are needed: energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms).The first quantum number describes the electron shell, or energy level, of an atom.
What is ML in quantum numbers?
Magnetic Quantum Number (ml): ml = -l, , 0, , +l. Specifies the orientation in space of an orbital of a given energy (n) and shape (l). This number divides the subshell into individual orbitals which hold the electrons; there are 2l+1 orbitals in each subshell.
2. Which quantum numbers gives the shell to which the electron belongs? Explanation: The principal quantum number, n, gives the shell to which the electron belongs.
Does aluminum have 13 valence electrons?
Aluminum atoms contain 13 electrons and 13 protons. There are 3 valence electrons in the outer shell. In standard conditions aluminum is a fairly soft, strong, and lightweight metal. Pure aluminum is a very reactive element and is rarely found on Earth in its free form.
How do you find the valence electrons of aluminium?
In the third shell, the number of valence electrons is = 3 electrons. Since [2+8+3=13] electrons, the atomic number of Aluminium is 13. In the second shell, the number of valence electrons = 6 electrons. Since [2+6=8] electrons, the atomic number of Oxygen is 8.
– The electronic configuration of the chlorine is [Ne]3s23p5 . – Therefore the quantum numbers for chlorine are principal quantum number (n) is 3, azimuthal quantum number (l) = 1 for 3p –orbital, magnetic quantum number (m) is -1, 0 , 1 and spin quantum (s) number is either −12or+12 .
Which set of quantum numbers are correct?
Hence the correct set of quantum numbers is: n=3, l=1, m=+1 or 0 or -1, s=+½ or -½. Conclusion: The correct set of quantum numbers among the given options is: 3, 1, -1, +½ i.e correct option is 3.
What is the quantum number for the last electron of iron Z 26?
The quantum number of 20th electron of Fe (Z = 26) ion would be 4, 0, 0, + 1/2.



