What 3 things happen during a combustion reaction?
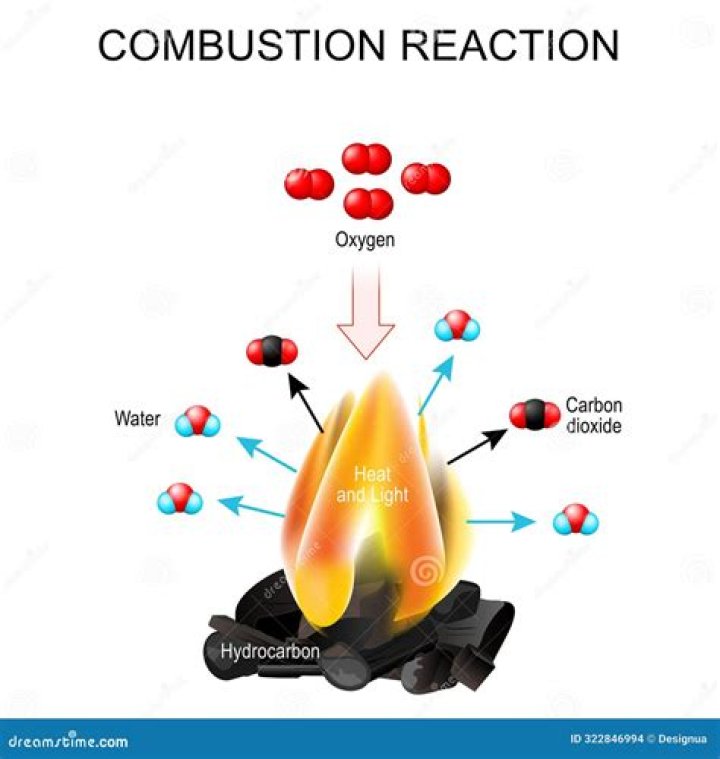
The second reactant is always a hydrocarbon, which is a compound made up of carbon and hydrogen. A combustion reaction also always produces CO2 and H2O.
What 3 things happen during a combustion reaction?
Combustion occurs and the match flames.
Three things are required in proper combination before ignition and combustion can take place—Heat, Oxygen and Fuel.
There must be Fuel to burn.There must be Air to supply oxygen.There must be Heat (ignition temperature) to start and continue the combustion process.
What is the best description of a combustion reaction?
Combustion is another name for burning. In a combustion reaction, fuel is burned and reacts with oxygen to release energy.
What is not true about combustion reactions?
What is not true about most combustion reactions? A carbon-based fuel is a reactant. The reaction has more than one product. MnSO4→MnO+SO3 is not a synthesis reaction.
Which reaction is a combustion reaction?
A combustion reaction (commonly known as burning ) is an exothermic reaction in which something reacts with oxygen. The combustion of organic compounds usually takes the form organic compound + oxygen => water + carbon dioxide.
What type of reaction is a combustion?
Combustion is a high-temperature exothermic (heat releasing) redox (oxygen adding) chemical reaction between a fuel and an oxidant, usually atmospheric oxygen, that produces oxidized, often gaseous products, in a mixture termed as smoke.
What is always a reactant in combustion reactions and what are the two types?
A combustion reaction always has oxygen as one reactant. The second reactant is always a hydrocarbon, which is a compound made up of carbon and hydrogen. A combustion reaction also always produces CO2 and H2O.
Why is combustion an exothermic reaction?
When a substance burns, it reacts with oxygen. This is known as combustion. All combustion reactions are exothermic because they release energy, eg heat energy is given out when methane is burned in a bunsen burner.
What are two products that are always formed in a combustion reaction?
A combustion reaction always has a hydrocarbon reacting with oxygen to produce carbon dioxide and water.
Which two products are always produced by a combustion reaction?
The two products from a complete combustion reaction of any fuel are carbon dioxide and water.
What is usually released in a combustion reaction quizlet?
What is usually released in a combustion reaction? Hydrogen, when combined with oxygen in a chemical reaction, forms water.
Is combustion always exothermic?
Combustion reactions are almost always exothermic (i.e., they give off heat). For example when wood burns, it must do so in the presence of O2 and a lot of heat is produced: When organic molecules combust the reaction products are carbon dioxide and water (as well as heat).
Is combustion an endothermic reaction?
Overall, combustion is an exothermic reaction given off or exiting , which means that energy is released. Energy is also needed for the fossil fuel to react with oxygen, so the energy use portion of combustion is an endothermic reaction into or absorbing .
What is combustion class8?
The process in which a substance undergoes a chemical reaction in the presence of air (oxygen) to produce heat and light is called combustion.
What is diffusion combustion?
In combustion, a diffusion flame is a flame in which the oxidizer and fuel are separated before burning. Diffusion flames tend to burn slower and to produce more soot than premixed flames because there may not be sufficient oxidizer for the reaction to go to completion, although there are some exceptions to the rule.
What is combustion system?
Combustion systems are chemical reactions between a material that can be burned as a fuel and the chemical compounds found in oxidants, substances that transfer oxygen atoms. The main purpose behind combustion is to create some sort of energy to produce the physical science definition of work.



