the free energy of activation is, check these out | What is the free energy of activation quizlet?
The free energies of activation and reaction are a measure of kinetics and thermodynamic favorability, respectively.
What is the free energy of activation quizlet?
The free energy of activation is the difference in energy between the substrate and the transition state.
What is free energy of activation in chemistry?
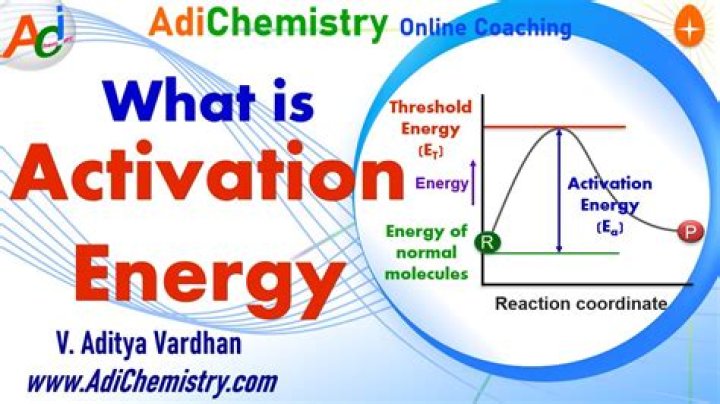
The activation energy for the forward reaction is the amount of free energy that must be added to go from the energy level of the reactants to the energy level of the transition state. The source of activation energy is typically heat, with reactant molecules absorbing thermal energy from their surroundings.
Is Gibbs free energy activation energy?
The free energy of a system changes during energy transfers, such as chemical reactions, and this change is referred to as ΔG or Gibbs free energy. All reactions require an input of energy called activation energy in order to reach the transition state at which they will proceed.
Is activation energy zero or negative?
Although the energy changes that result from a reaction can be positive, negative or even zero, before a reaction can occur, an energy barrier must be resolved in both situations. This means the energy for activation is still positive. In certain cases, with increasing temperature, reaction rates decrease.
How does Gibbs free energy relate to equilibrium?
ΔG is related to Q by the equation ΔG=RTlnQK. If ΔG Q, and the reaction must proceed to the right to reach equilibrium. If ΔG > 0, then K
What can be said about the Gibbs free energy in an exergonic reaction quizlet?
During a spontaneous change, free energy decreases and the stability of a system increases. In an exergonic reaction, energy is released to the surroundings. The bonds being formed are stronger than the bonds being broken. In an endergonic reaction, energy is absorbed from the surroundings.
What is free energy profile?
Free energy profile (FE Profile) is an essential quantity for the estimation of reaction rate and the validation of reaction mechanism. This method can significantly enhance the applicability of ai QM/MM methods in the studies of chemical reactions in condensed phase and enzymatic reactions.
What is activation energy Ncert?
The minimum quantitiy of external energy required for the conversion of reactant into product or to produce an unstable intermediate is called activation energy. It is E. Rate of reaction is inversely proportional to the activation energy.
What is free energy in enzyme?
a catalyzed chemical reaction. Gibbs Free Energy (G) is used to describe the useful energy in a reaction or the energy capable of doing work. Enzymes do affect the activation energy. The activation energy is the difference in free energy between the substrate and the transition state.
What is enzyme activation energy?
Lesson Summary. The activation energy is the energy required to start a reaction. Enzymes are proteins that bind to a molecule, or substrate, to modify it and lower the energy required to make it react.
What represents the activation energy?
activation energy, in chemistry, the minimum amount of energy that is required to activate atoms or molecules to a condition in which they can undergo chemical transformation or physical transport.
Is activation energy positive or negative?
Activation energy is typically positive. It’s the minimum energy required for a chemical reaction to proceed. There are few other ways to describe the activation energy but they basically say the same thing.
Is activation energy can be zero?
We can say activation energy as the minimum possible quantity of energy (minimum) that is necessary to initiate a reaction or the quantity of energy existing in a chemical system for a reaction to occur. Thus, a chemical reaction could not have zero energy of activation.
Is the activation energy always positive?
This means that the activation energy is almost always positive; there is a class of reactions called barrierless reactions, but those are discussed elsewhere. For similar reactions under comparable conditions, the one with the smallest Ea will occur most rapidly.
What does Gibbs free energy tell us?
Gibbs free energy measures the useful work obtainable from a thermodynamic system at a constant temperature and pressure. Just as in mechanics, where potential energy is defined as capacity to do work, similarly different potentials have different meanings.
What is free energy of a reaction quizlet?
Free energy. the total amount of energy in a system (a cell) that can be tapped to do work. Not all energy transfers are 100% Exergonic Reactions.
Why is Gibbs free energy at equilibrium?
Both the forward and backward reactions occur at the same rate. If the Gibbs energy change for the forward reaction is G, then the Gibbs energy change is -G for the backward reaction. Total Gibbs energy at equilibrium is G+(-G)=0. This is why Gibbs energy is zero at equilibrium.



