purpose of immunoprecipitation, check these out | What is the principle of immunoprecipitation?
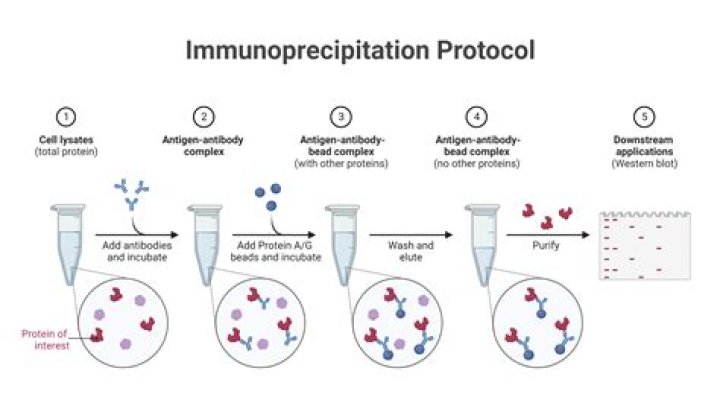
Immunoprecipitation (IP) is the technique of precipitating a protein antigen out of solution using an antibody that specifically binds to that particular protein. This process can be used to isolate and concentrate a particular protein from a sample containing many thousands of different proteins.
What is the principle of immunoprecipitation?
Immunoprecipitation (IP) is a method to isolate a specific antigen from a mixture, using the antigen-antibody interaction. Antigens isolated by IP are analyzed by SDS-PAGE or Western blotting.
What are advantages of immunoprecipitation?
Immunoprecipitation has an advantage in that the antigens are allowed to react with the antibodies in their native conformation prior to their subsequent separation and quantification. A further advantage is that a protein at a very low concentration can be concentrated from the relatively large volume of 1–2 mL.
What is immunoprecipitation technique?
Immunoprecipitation (IP) is the small-scale affinity purification of antigens using a specific antibody that is immobilized to a solid support such as magnetic particles or agarose resin.
What is the difference between Western blot and immunoprecipitation?
Immunoprecipitation involves using antibodies and agarose beads to isolate a target protein from a solution, while western blotting (also known as immunoblotting) uses gel electrophoresis and an antibody probe to analyze proteins.
Who discovered immunoprecipitation?
History and New ChIP methods. In 1984 John T. Lis and David Gilmour, at the time a graduate student in the Lis lab, used UV irradiation, a zero-length protein-nucleic acid crosslinking agent, to covalently cross-link proteins bound to DNA in living bacterial cells.
Is Elisa an immunoprecipitation?
Immunoprecipitation is a technique in which an antigen is isolated by binding to a specific antibody attached to a sedimentable matrix. Performing an ELISA involves at least one antibody with specificity for a particular antigen.
How is immunoelectrophoresis performed?
Immunoelectrophoresis is performed by placing serum on a slide containing a gel designed specifically for the test. An electric current is then passed through the gel, and immunoglobulins, which contain an electric charge, migrate through the gel according to the difference in their individual electric charges.
Is immunoprecipitation quantitative?
Unlike column affinity chromatography, the goal of immunoprecipitation is to isolate just enough protein to be able to measure it by western blotting or other semi-quantitative or quantitative assay methods. Usually treated and untreated samples are compared to assess the relative amount of the protein of interest.
What is the purpose of immunofluorescence?
Immunofluorescence (IF) is an important immunochemical technique that allows detection and localization of a wide variety of antigens in different types of tissues of various cell preparations.
Why is it called immunoprecipitation?
The beads with bound antibodies are then added to the protein mixture, and the proteins that are targeted by the antibodies are captured onto the beads via the antibodies; in other words, they become immunoprecipitated.
How do you analyze immunoprecipitation?
Analysis of the immunoprecipitate is usually by electrophoresis although other techniques can be used. The choice of immobilized antibody binding protein depends upon the species that the antibody was raised in.
Can you IP and blot with same antibody?
The proprietary Detection Reagent does not recognize denatured heavy or light chains from monoclonal antibodies; therefore, the same monoclonal antibody can be used for the IP step and post-IP Western blot detection.
What is the difference between IP and co-IP?
Difference between IP and co-IP is the focus of the experiment. IP is focused on the primary target, which binds the antibody. Whereas, Co-IP targets the secondary targets, which interacts with the primary proteins, instead of antibody.
When was immunoprecipitation created?
Chromatin immunoprecipitation (ChIP), originally developed by John T. Lis and David Gilmour in 1984, has been useful to detect DNA sequences where protein(s) of interest bind.
What is immunoprecipitation in immunology?
Immunoprecipitation is a technique in which an antigen is isolated by binding to a specific antibody attached to a sedimentable matrix. It is also used to analyze protein fractions separated by other biochemical techniques such as gel filtration or density gradient sedimentation.



