poly functional group, check these out | What are polyfunctional compounds and examples?
Organic compounds which contain two or more functional group are called polyfunctional compounds.
What are polyfunctional compounds and examples?
An example for poly-functional compound is 4-nitro aniline. It contains nitro group and aminio groups as functional groups. Other examples includes glycine, glucose, salicylic acid etc. A poly-functional compound has more than one functional group.
What makes a compound polyfunctional?
feature of a monomer is polyfunctionality, the capacity to form chemical bonds to at least two other monomer molecules. Bifunctional monomers can form only linear, chainlike polymers, but monomers of higher functionality yield cross-linked, network polymeric products.
What are functional groups?
A functional group is a group of atoms in a molecule with distinctive chemical properties, regardless of the other atoms in the molecule. The atoms in a functional group are linked to each other and to the rest of the molecule by covalent bonds.
What is a monofunctional compound?
[‚män·ō¦fəŋk·shən·əl ′käm‚pau̇nd] (organic chemistry) An organic compound whose chemical structure possesses a single highly reactive site.
What is poly functional?
: having many functions polyfunctional acids.
What compounds are called polyfunctional and Heterofunctional?
Dicarboxylic acids, such as oxalic, malonic, succinic, have been discussed in the preceding chapter. These compounds are referred to as polyfunctional ones. A significant importance in living systems belongs to heterofunctional compounds that involve different functional groups in the same molecule.
What is the aldehyde group?
aldehyde, any of a class of organic compounds in which a carbon atom shares a double bond with an oxygen atom, a single bond with a hydrogen atom, and a single bond with another atom or group of atoms (designated R in general chemical formulas and structure diagrams).
What are carboxylic acids?
carboxylic acid, any of a class of organic compounds in which a carbon (C) atom is bonded to an oxygen (O) atom by a double bond and to a hydroxyl group (―OH) by a single bond. A fourth bond links the carbon atom to a hydrogen (H) atom or to some other univalent combining group.
How do you name organic compounds with multiple functional groups?
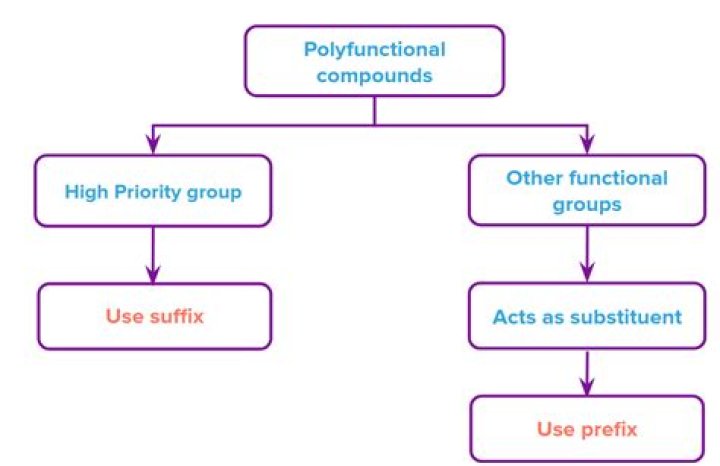
If the compound includes more than one functional groups, the one with the highest priority is the “parent structure” and determines the “parent name”; the other groups will be regarded as “substituents”. “Suffix” is used to indicate the name of the parent structure, and “prefix” is for the substituent.
What are the 7 functional groups?
Functional groups include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl.
What are the 4 functional groups?
What are the four functional groups? In biological molecules, some of the essential functional groups include hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl groups. These groups play a significant role in forming molecules such as DNA, proteins, carbohydrates, and lipids.
What are the 8 functional groups?
Hydroxyl, sulfhydryl, carbonyl, carboxyl, amino and phosphate groups. Alcohols and thiols.
What is monofunctional group?
Adjective. monofunctional (not comparable) Having a single function. (organic chemistry) Having a single functional group.
What is monofunctional example?
: of, relating to, or being a compound with one reactive site in a molecule (as in polymerization) formaldehyde is a monofunctional reagent.
What is monomeric unit?
A monomer is a molecule that forms the basic unit for polymers, which are the building blocks of proteins. Monomers bind to other monomers to form repeating chain molecules through a process known as polymerization. Monomers may be either natural or synthetic in origin.
How are organic compounds classified according to solubility?
Organic compounds follow three interdependent rules of solubility: 1. small organic molecules are more soluble in water than are large organic molecules; 2. polar organic molecules, especially those capable of hydrogen bonding, are more soluble in water than are nonpolar molecules; and 3.
Which is trifunctional monomer?
The simple molecules that may become structural units are themselves called monomers; two monomers combine to form a dimer, and three monomers, a trimer. When the structural unit is trifunctional (has three bonding sites), a nonlinear, or branched, polymer results.
What is bifunctional monomer?
Bifunctional polymer means which has two functional groups in its every monomer and trifunctional polymer means the one having three functional groups in its every monomer.



