maxwell boltzmann distribution curve, check these out | What is Maxwell-Boltzmann distribution law?
What is Maxwell-Boltzmann distribution law?
This law states that the average amount of energy involved in each different direction of motion of an atom is the same. He derived an equation for the change of the distribution of energy among atoms due to atomic collisions and laid the foundations of statistical mechanics.
What does a Boltzmann distribution show?
The Boltzmann distribution (AKA kinetic energy distribution) is a distribution that describes the amount of kinetic energy that a proportion of gas particles have in a given sample.
Why is the Boltzmann distribution important?
The Boltzmann distribution gives the probability that a system will be in a certain state as a function of that state’s energy, while the Maxwell-Boltzmann distributions give the probabilities of particle speeds or energies in ideal gases.
How does molar mass affect Maxwell-Boltzmann distribution?
Figure 3 shows the dependence of the Maxwell-Boltzmann distribution on molecule mass. On average, heavier molecules move more slowly than lighter molecules. Therefore, heavier molecules will have a smaller speed distribution, while lighter molecules will have a speed distribution that is more spread out.
What is Boltzmann distribution chemistry?
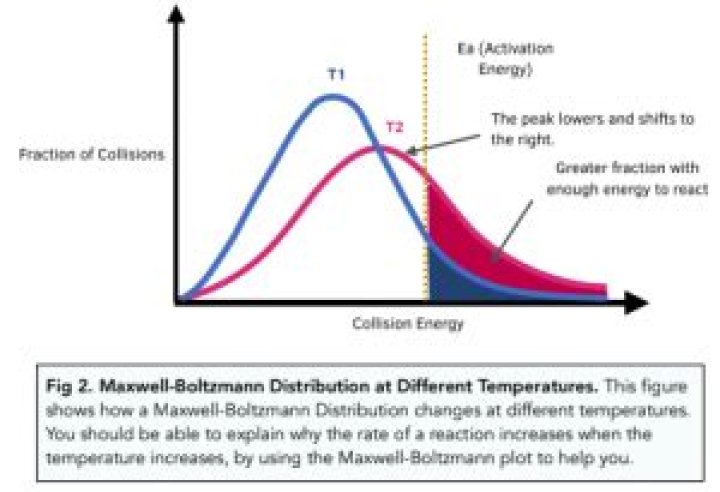
A Boltzmann Distribution shows the distribution of molecular energies in a gas at constant temperature. Most gas molecules have energies within a comparatively narrow range. • The curve will only meet the energy axis at infinity energy. No molecules have zero energy.
What is Boltzmann distribution equation?
In these equations, n = ∫ 0 ∞ f ( ε ) d ε is the number density, T is the temperature of electrons, Γ is the gamma function, and kB is the Boltzmann constant.
Is the Maxwell-Boltzmann curve symmetrical?
Maxwell-Boltzmann Distributions
This results in an asymmetric curve, known as the Maxwell-Boltzmann distribution. The peak of the curve represents the most probable velocity among a collection of gas particles. Velocity distributions are dependent on the temperature and mass of the particles.
Does the Maxwell-Boltzmann distribution only apply to gases?
So the answer to the question asked in your title, “Does the Maxwell-Boltzmann distribution apply to gases only?” is “no”; it applies to all phases of matter, in the sense that it describes the distribution of particle speeds and energies.



