list the three subatomic particles, check these out | What are the 3 subatomic particles?
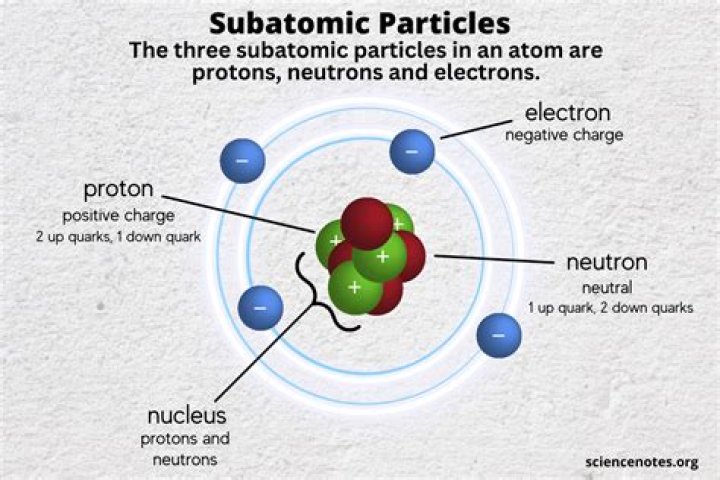
There are three subatomic particles: protons, neutrons and electrons. Two of the subatomic particles have electrical charges: protons have a positive charge while electrons have a negative charge.
What are the 3 subatomic particles?
Given that these particles make up atoms, they are often referred to as subatomic particles. There are three subatomic particles: protons, neutrons and electrons. Two of the subatomic particles have electrical charges: protons have a positive charge while electrons have a negative charge.
What are the 3 subatomic particles quizlet?
The three subatomic particles are protons, neutrons, and electrons.
What are the 3 subatomic particles their charges and location?
Proton (charge of +e, in the nucleus), Neutron (0 charge, in the nucleus), and Electron (charge of –e, outside the nucleus).
What are the types of subatomic particles?
A typical atom consists of three subatomic particles: protons, neutrons, and electrons (as seen in the helium atom below). Other particles exist as well, such as alpha and beta particles (which are discussed below). The Bohr model shows the three basic subatomic particles in a simple manner.
What is the most important subatomic particle?
Electrons. The electron is one of the most important subatomic particle, it is small compared to protons and neutrons.
What is a subatomic particle called?
subatomic particle, also called elementary particle, any of various self-contained units of matter or energy that are the fundamental constituents of all matter.
What are three types of charges?
Electric Charge
protons are positively charged.electrons are negatively charged.neutrons have zero charge.
How do you find subatomic particles?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
What are the 3 types of electrons?
There are charged leptons and uncharged leptons. Electron, Muon and Tau are the charged leptons. And their associated neutrinos are the uncharged leptons. The only difference between the 3 charged leptons is their mass, with each successive lepton being heavier than previous one.
Do atoms have 3 main parts and 2 subatomic particles?
Atoms are composed of three main subatomic particles: protons, neutrons, and electrons. Protons and neutrons are grouped together in the nucleus of an atom, while electrons orbit about the nucleus.
What are the subatomic particles of an atom Class 9?
There are three subatomic particles, which are namely electrons, protons and neutrons.



