Is water polar or nonpolar?
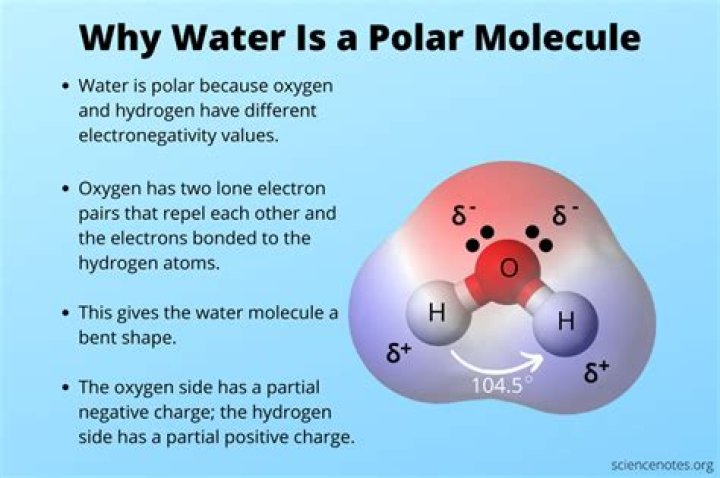
Water is a polar molecule. While the overall charge of the molecule is neutral, the orientation of the two positively charged hydrogens (+1 each) at one end and the negatively charged oxygen (-2) at the other end give it two poles.
Is water polar or nonpolar and why?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.
Why is water called polar?
Unequal sharing of electrons makes water a polar molecule. This makes the oxygen end of the molecule slightly negative. Since the electrons are not near the hydrogen end as much, that end is slightly positive. When a covalently bonded molecule has more electrons in one area than another, it is called a polar molecule.
Water is a Polar Covalent Molecule
Water (H2O), like hydrogen fluoride (HF), is a polar covalent molecule. When you look at a diagram of water (see Fig. 3-2), you can see that the two hydrogen atoms are not evenly distributed around the oxygen atom.
Is water hydrophobic?
The textbooks say that water readily comes together with other water, open arms of hydrogen clasping oxygen attached to other OH molecules. “In other words, the first layer of water is hydrophobic.”
Water is also a kind of skeleton. It consists of tiny particles, the atoms, just like every other substance on earth. One of these atoms is called hydrogen and the other is called oxygen. One particle of water is called a molecule.
Is water a molecular?
The key to understanding water’s chemical behavior is its molecular structure. A water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons.
Why is water cohesive?
Water is highly cohesive—it is the highest of the non-metallic liquids. More precisely, the positive and negative charges of the hydrogen and oxygen atoms that make up water molecules makes them attracted to each other.
Water isn’t wet because it is a liquid that wets things. Once you come into contact with water you become wet. Until then water is liquid and you are dry.
Is water wet a paradox?
Is water wet? Answer 1: Liquid water is not itself wet, but can make other solid materials wet. Wetness is the ability of a liquid to adhere to the surface of a solid, so when we say that something is wet, we mean that the liquid is sticking to the surface of a material.
Can water wet water?
According to information obtained from a scholarly database, Quora.com, “In a liquid-liquid interaction, such as water by itself, we can say that water is not wet, as molecules are all bound together and not wetting one another.” Though water has the ability to make other materials wet, the liquid itself is not wet.
What is the geometry of water?
The molecular geometry of the water molecule is bent. The H-O-H bond angle is 104.5°, which is smaller than the bond angle in NH3 (see Figure 11). Figure 11. Water molecule.
Etymology. The word water comes from Old English wæter, from Proto-Germanic *watar (source also of Old Saxon watar, Old Frisian wetir, Dutch water, Old High German wazzar, German Wasser, vatn, Gothic ���� (wato), from Proto-Indo-European *wod-or, suffixed form of root *wed- (“water”; “wet”).
Why does water have a bent geometry?
The oxygen has 6 valence electrons and thus needs 2 more electrons from 2 hydrogen atoms to complete its octet. The water molecule is bent molecular geometry because the lone electron pairs, although still exerting influence on the shape, are invisible when looking at molecular geometry.



