Is Vinegar a polar molecule?
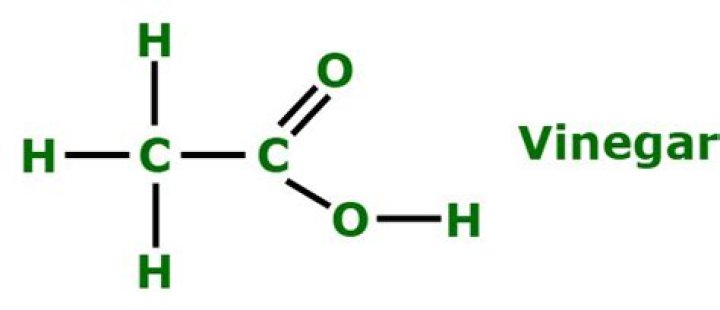
Vinegar is composed of acetic acid and water, which are polar compounds. In a polar molecule, one or a group of atoms have a stronger pull on the electrons in the molecule. … Therefore, nonpolar molecules do not have such well-developed dipoles.
Is water and vinegar polar?
Water, acetic acid, and alcohol are all examples of polar molecules—molecules that have a slightly negative charge at one end, or pole, and a slightly positive charge at another end.
Which is a polar molecule?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A diatomic molecule that consists of a polar covalent bond, such as HF, is a polar molecule. A molecule with two poles is called a dipole (see Figure below ) . Hydrogen fluoride is a dipole.
For example, the molecular formula for acetic acid, the component that gives vinegar its sharp taste, is C2H4O2. This formula indicates that a molecule of acetic acid (Figure 2.21) contains two carbon atoms, four hydrogen atoms, and two oxygen atoms. The ratio of atoms is 2:4:2.
Is acetic anhydride polar?
Acetic Anhydride has a non-polar structure along with flexible molecules. Molecular Formula is C4H6O3. Molar Mass is 102.09 g/mol.
If the arrangement is symmetrical and the arrows are of equal length, the molecule is nonpolar.If the arrows are of different lengths, and if they do not balance each other, the molecule is polar.If the arrangement is asymmetrical, the molecule is polar.
Is vinegar a polar nonpolar or ionic?
As discussed earlier, vinegar is a polar solution of acetic acid and water, with a few other compounds present in varying amounts which produce the flavor nuances that make different vinegars unique.
Is vinegar soluble?
The product vinegar is hydrophilic in nature, which means it likes water. As a result, it doesn’t dissolve in water but absorbs water on a molecular level, giving the appearance of a soluble solution.
By definition, solutions are uniform/homogeneous mixtures. Examples would be Kool-Aid, fruit punch, salt water, vinegar, vodka, etc. On the other hand, non-uniform mixtures are also called heterogeneous mixtures.
What makes a molecule polar covalent?
A polar covalent bond is a covalent bond in which the atoms have an unequal attraction for electrons and so the sharing is unequal. In a polar covalent bond, sometimes simply called a polar bond, the distribution of electrons around the molecule is no longer symmetrical.
What is meant by non polar molecules?
A nonpolar molecule is one whose charge distribution is spherically symmetric when averaged over time; since the charges oscillate, a temporary dipole moment exists at any given instant in a so-called nonpolar molecule.
What are examples of a polar covalent bond?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.
A polar molecule forms when an atom of high electronegativity (one that attracts electrons), such as chlorine, bonds with a less electronegative atom such as hydrogen. Polar molecules tend to align themselves because the negative end of each molecule is attracted to the positive end of other molecules, and vice versa.
Are all asymmetrical molecules polar?
Asymmetrical molecules are always polar. Symmetrical molecules can be polar and non-polar. Polarity is dependent on the difference in electronegativity of the molecule’s atoms and shape. The atom with the largest electronegativity will attract the electrons more frequently than the other atoms in the molecule.



