Is SOCl2 trigonal planar?
The molecular geometry of SOCl2 is trigonal pyramidal and its electron geometry is tetrahedral.
Is SOCl2 trigonal planar?
SOCl2 is trigonal pyramidal, and is polar. There is a lone pair of electrons on S as well as three bonds (the bond to O is a double bond). Since it is trigonal pyramidal it is asymmetrical and the vector sum of the bond dipoles is nonzero.
What is the electron pair geometry for S in SO2Cl2?
The molecular geometry of SO2Cl2 is tetrahedral with an asymmetric charge distribution on the central atom. Therefore this molecule is polar. Beside above, what is the hybridization of SO2Cl2? Hybridization of S The hybridization that gives a tetrahedral structure is sp3 .
Is SOCl2 T shaped?
The answer is d.
The structure of thionyl chloride showing the trigonal pyramidal geometry is shown below.
Why is there a double bond in SOCl2?
Originally Answered: Why is SOCl2 composed with a double bond? S=O is a double bond. This above is supposed to describe 2 Chlorine atoms each attached by a single bond to the Sulfur atom, with a double bond attaching the Oxygen molecule. This is thionyl chloride, a very reactive chemical compound.
What is the molecular geometry of ph3?
In essence, ph3 is a Drago molecule and if we look at its bond angle data it shows that the p-orbitals have an angle of 90掳. Looking at its Lewis structure we can state that molecular geometry of PH3 is trigonal pyramidal.
Is so2 molecule bent?
In sulphur dioxide, as well as the two double bonds, there is also a lone pair on the sulphur. To minimise repulsions, the double bonds and the lone pair get as far apart as possible, and so the molecule is bent.
What is the electron-pair geometry for S in SO2Cl2 fill in the blank 4 there are lone pair’s around the central atom so the geometry of SO2Cl2 is?
The Lewis diagram for SO2Cl2 is: The electron-pair geometry around the S atom in SO2Cl2 is fill in the blank 4. There are lone pair(s) around the central atom, so the geometry of SO2Cl2 is. Alkendra S.
What is structure of SO2?
Sulfur dioxide (SO2) is a bent or V-shape molecule with its bond order of 1.5. It has S鈭扥鈭扴 bond angle 119 and bond dissociation enthalpy is 297kJ/mol. The 蟻 bonds between S and O are formed by sp2-p overlapping. Sulfur in (SO2)is sp2 hybridised forming three hybrid orbitals.
What is the molecular geometry of PF6?
The VSEPR model for [PF6] – is Octahedral.
What molecules have linear geometry?
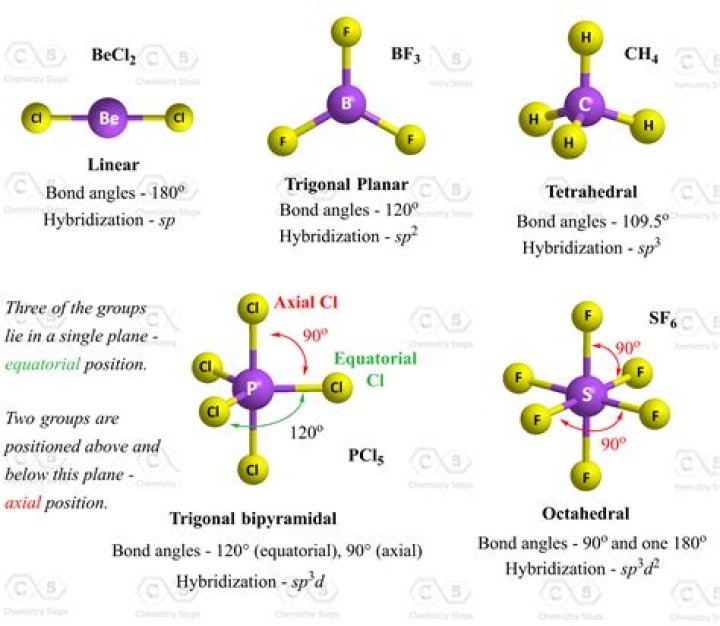
Linear molecule is a molecule in which atoms are deployed in a straight line (under 180掳 angle). Molecules with an linear electron pair geometries have sp hybridization at the central atom. An example of linear electron pair and molecular geometry are carbon dioxide (O=C=O) and beryllium hydride BeH2.
Which is trigonal pyramidal?
Trigonal pyramidal is a molecular shape that results when there are three bonds and one lone pair on the central atom in the molecule. Molecules with an tetrahedral electron pair geometries have sp3 hybridization at the central atom. Ammonia (NH3) is a trigonal pyramidal molecule.



