Is SF4 seesaw shape?
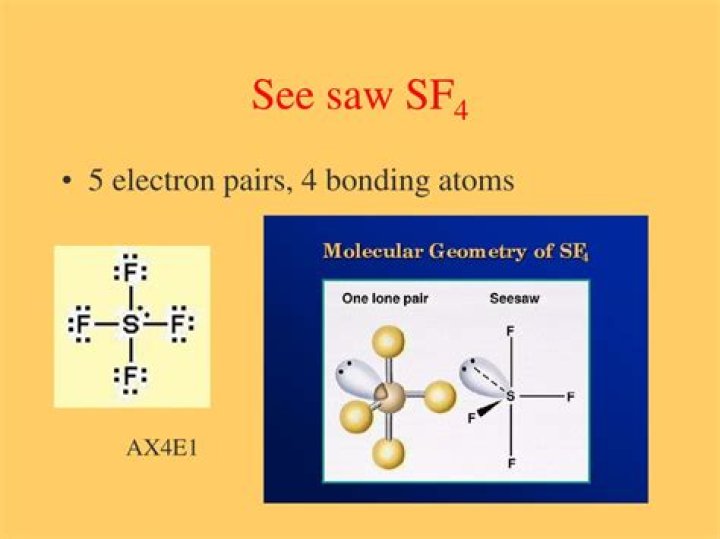
An example of a seesaw shaped molecule is sulfur tetrafluoride, or SF4. Sulfur is the central atom, two fluorine atoms are on the equatorial plane, and two are on the axial plane.
Why SF4 has a seesaw shape?
This shape is caused by a lone pair of electrons on the central atom. An example of a seesaw shaped molecule is sulfur tetrafluoride, or SF4. The equatorial plane fluorine atoms form an angle of 89 degrees, and the angle formed from the equatorial plane fluorine atoms to the axial plane fluorine atoms is 103 degrees.
Is SF4 bent or linear?
There are various types of Molecular structures such as linear, tetrahedral, bent, octahedral, trigonal pyramidal, trigonal planar, and more. SF4 covers under the ‘Trigonal Bipyramidal’ structure because of its electron arrangements.
The Lewis structure for SF4 contains four single bonds and a lone pair on the sulfur atom. Because of the greater repulsion of a lone pair, it is one of the equatorial atoms that are replaced by a lone pair. The geometry of the molecule is called a distorted tetrahedron or seesaw.
Is SF4 seesaw or trigonal bipyramidal?
SF4 Sulfur Tetrafluoride
These are arranged in a trigonal bipyramidal shape with 102° F-S-F bond angles between the equatorial fluorine atoms and 173° between the axial fluorine atoms. The lone pair takes an equatorial position because it demands more space than the bonds.
It has a square planar geometry.
Is SF4 polar or nonpolar molecule?
The Sulfur tetrafluoride is a polar molecule because Fluorine is more electronegative than Sulfur. With this, the distribution of the charge is not equal, making the SF4 polar molecules. The molecular geometry of SF4 is in a seesaw molecular shape that can be seen when you draw the Lewis Structure.
What is the preferred geometry of SF4?
The electrons follow this pattern of arrangement following the VSEPR rule to minimize the repulsion forces between the lone pairs of electrons to maximize the molecule’s stability. Hence, SF4 has a trigonal bipyramidal molecular geometry.
SF4 (Sulfur tetrafluoride) is polar in nature as sulfur atom consists of a lone pair on it due to which the shape of the molecule becomes asymmetric ie; seesaw.
Is SF4 a molecular solid?
Molecular solid: composed of covalent bonds. SF4: This compound is composed of S (a nonmetal) and F (a nonmetal) connected by a covalent bond. This compound is molecular.
What is the Vsepr type of SF4 with a seesaw molecular geometry?
SF4 molecular geometry is see-saw with one pair of valence electrons. The nature of the molecule is polar. These atoms form a trigonal bipyramidal shape.
Does SF4 obey octet rule?
Lewis Dot of Sulfur Tetrafluoride SF4. S does not follow the octet rule. It will hold more than 8 electrons. Sulfur having valence electrons in the 3rd energy level, will also have access to the 3d sublevel, thus allowing for more than 8 electrons.
Disphenoidal or Seesaw is a type of molecular geometry where there are four bonds to a central atom with overall C2v molecular symmetry. Most commonly, four bonds to a central atom result in tetrahedral or, less commonly, square planar geometry.
What is the molecular geometry of an SF4 molecule?
The molecular geometry of SF4 according to its molecular formula and hybridization is trigonal bipyramidal. The shape generally resembles a see-saw and this shape is due to the repulsion in bonding and lone pairs of electrons.



