Is PH3 London dispersion?
It forms dipole-dipole because it is a polar molecule. Here is why: PH3 is called phosphine and it is quite toxic and flammable. PH3 must be polar since it is not symmetrical.
Is PH3 London dispersion?
Answer Both phosphine (PH3) and ammonia (NH3) have London dispersion forces. Phosphine with a molecular mass of 34g/mole is a larger and heavier molecule than ammonia with its molecular mass of 17g/mole. Hence, phosphine has stronger London dispersion forces.
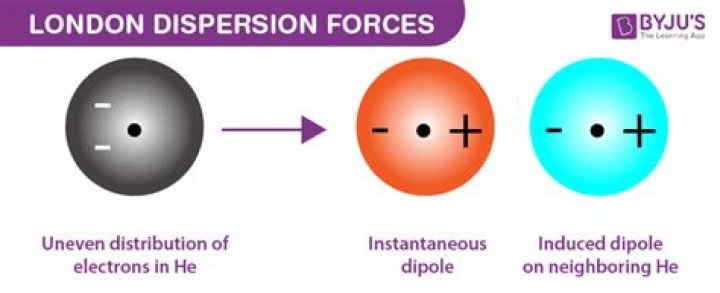
Does PH3 form dispersion forces?
PH3 forms dispersion forces, and NH3 does not.
Is NH3 dipole-dipole?
Why NH3 is called dipole-dipole? You know that, ammonia is a polar molecules. it exhibits, dipole-dipole intraction, induced attraction, and London dispersion forces. NH3 is called dipole dipole because nh3 make N-H bond, it directly make hydrogen bonding.
Is SO2 dipole-dipole?
Yes, sulfur dioxide (SO2) is a polar molecule that feature dipole dipole interactions in its intermolecular forces.
What type of bond is PH3?
The electronegative property of PH3 found in the periodic table attracts shared pairs of valence electrons, creating covalent bonds. However, due to the unbonded electron, there will be asymmetrical charge distribution. With this, the PH3 is a polar molecule with nonpolar covalent bonds, not polar bonds.
Is ch4 dipole-dipole?
Because methane is a non-polar molecule it is not capable of hydrogen bonding or dipole-dipole intermolecular forces. The electronegativities of C and H are so close that C-H bonds are nonpolar. There are no bond dipoles and no dipole-dipole interactions.
How do you tell if a molecule has a dipole?
A dipole exists when a molecule has areas of asymmetrical positive and negative charge. A molecule’s polarity (its dipole) can be experimentally determined by measuring the dielectric constant. Molecular geometry is crucial when working with dipoles.
Which molecule is a dipole?
Molecules that contain dipoles are called polar molecules and are very abundant in nature. For example, a water molecule (H2O) has a large permanent electric dipole moment.
What intermolecular force is Br2?
Br2 B r 2 exhibits only London dispersion forces. This is because diatomic elements are nonpolar and nonpolar molecules are only capable of
Is HF dipole-dipole or dispersion?
HF is a polar molecule: dipole-dipole forces. Hydrogen is bounded to F. Hydrogen bonds exist. There are also dispersion forces between HBr molecules.
What is the intermolecular force of ch3cooh?
In acetic acid (CH3COOH), hydrogen bonding, dipole-dipole interactions and dispersion force are present whereas in carbon tetrachloride (CCl4) only dispersion non-polar forces are present.
Is Br2 dipole-dipole?
Thus, the boiling points will increase as the London Forces increase and this is shown in the following table. bromine Br2 114 −7 liquid iodine I2 133 114 solid The intermolecular forces (dipole-dipole, hydrogen bonding and London) are all a form of dipole- dipole, whether temporary or permanent.
Is H2 dipole-dipole?
If the molecules have no dipole moment, (e.g., H2, noble gases etc.) then the only interaction between them will be the weak London dispersion (induced dipole) force.
What type of intermolecular force is H2CO?
H2CO is a polar molecule and will have both dipole-dipole forces and London dispersion forces while CH3CH3 is a non-polar molecule and will only have London dispersions forces.
How polar is SO2?
SO2 is polar in nature because of the difference in electronegativity between sulfur and oxygen atoms. The greater the difference in electronegativity more will be the polarity of the molecule. The bent shape of SO2 is because of the repulsion between the unbonded electrons present on the sulfur and oxygen atoms.
Is SO2 a hydrogen bond?
SO2 can’t form hydrogen bonds because there is no hydrogen. The bonds are polar as the S is slightly positive and the O is slightly negative, so there is some intermolecular bonding happening there, but without hydrogen, they are not classified as hydrogen bonds.



