Is PCl3 polar or nonpolar?
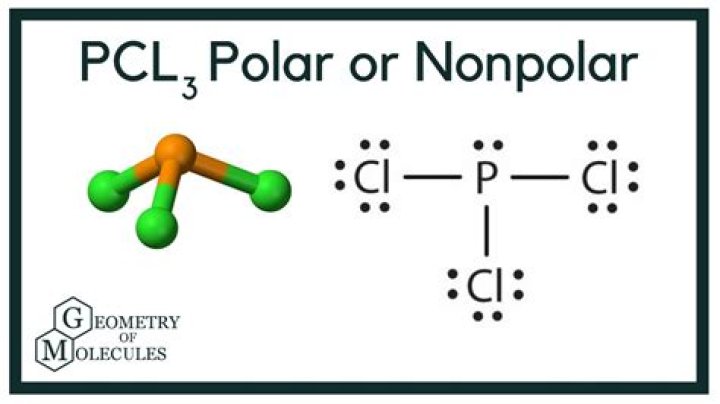
PCl3 is a polar molecule because of its tetrahedral geometrical shape having a lone pair on Phosphorus atom and the difference between the electronegativity of Chlorine(3.16) and Phosphorus(2.19) atoms resulting in unequal sharing of electrons and develop positive and negative poles across the molecule making it a …
What kind of bond is PCl3?
PCl3 is a polar molecule because of its geometry and difference in electronegativity between the 2 atoms. Again another question like, whether PCl3 is ionic or covalent, can pop up in your mind. So, PCl3 is a covalent molecule because here equal sharing of electrons forms the bond between phosphorus and chlorine.
Is it polar or non polar?
When things are different at each end, we call them polar. Some molecules have positive and negative ends too, and when they do, we call them polar. If they don’t, we call them non-polar. Things that are polar can attract and repel each other (opposite charges attract, alike charges repel).
The phosphorus atom in the covalent compound phosphorus trichloride, PCl3 , has an electronegativity of 2.19, and the chlorine atoms each have an electronegativity of 3.16, therefore the difference in electronegativity is 0.97, indicating a polar covalent bond between the phosphorus atom and each of the three chlorine
What is molecular geometry of PCl3?
Looking at the PCl3 molecular geometry it is trigonal pyramidal with a bond angle of approx. 103o. The is mainly due to the disproportionate influence or greater repulsion of the phosphorus lone pair which makes it deviate from the ideal angle of 109o.
(A) PCl3 Is contains three covalent bond which are formed by equal sharing of electron in between phosphorus atom and chlorine atom, hence it’s a covalent molecule.
Which molecule is nonpolar?
Nonpolar Molecule Examples
Examples of homonuclear nonpolar molecules are oxygen (O2), nitrogen (N2), and ozone (O3). Other nonpolar molecules include carbon dioxide (CO2) and the organic molecules methane (CH4), toluene, and gasoline. Most carbon compounds are nonpolar.
What electronegativity is nonpolar?
Although there are no hard and fast rules, the general rule is if the difference in electronegativities is less than about 0.4, the bond is considered nonpolar; if the difference is greater than 0.4, the bond is considered polar.
Any of the homonuclear diatomic elements: H2, N2, O2, Cl2 (These are truly nonpolar molecules.) Carbon dioxide – CO. Benzene – C6H. Carbon tetrachloride – CCl.
Is PCl3 a planar molecule?
No. PCl3 is not trigonal planar. It is trigonal pyramidal in shape.
Does PCl3 have dipole dipole forces?
PCl3 is a polar molecule and its strongest intermolecular forces are dipole-dipole interactions. It has the next highest melting point.
What is the 3d shape of PCl3?
The molecular geometry of PCl3 is trigonal pyramidal with asymmetric charge distribution on the central atom. Therefore this molecule is polar.
Explanation & Answer
Phosphorus trichloride has a lone pair, and therefore can act as a Lewis base. For example with the Lewis acids BBr3 it forms a 1:1 adduct, Br3B−−+PCl3.
Is PCl3 good conductor of heat or electricity?
Is pcl3 a good conductor of heat or electricity? Phosphorus(III) chloride (PCl3) The liquid does not conduct electricity because of the lack of ions or mobile electrons.



