Is O2 polar or nonpolar?
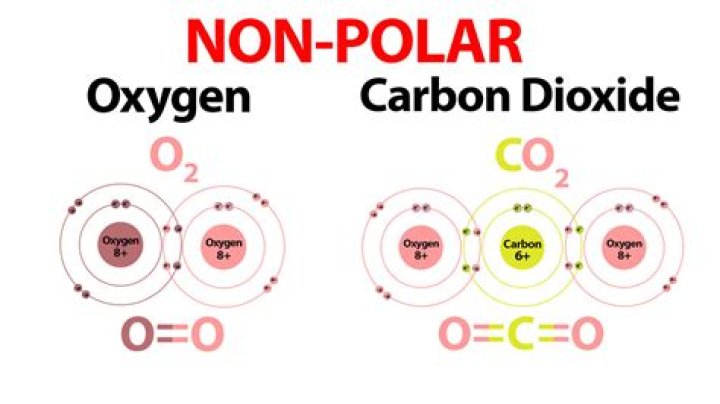
For example, molecular oxygen (O2) is nonpolar because the electrons will be equally distributed between the two oxygen atoms. Another example of a nonpolar covalent bond is methane (CH4), also shown in Figure 1.
Is 02 a non-polar covalent bond?
A nonpolar covalent bond occurs when atoms share electrons equally, and the electrons do not spend more time around either of the atoms. An oxygen gas (O2) molecule has a nonpolar covalent bond.
Is O2 hydrophobic or hydrophilic?
Nonpolar molecules, such as hydrocarbons, CO2 and O2, are hydrophobic.
O2 is a covalent molecule because each oxygen atom needs two valence electrons to complete its octet. To meet this need, each oxygen atom shares two of its electrons with the other oxygen forming a strong oxygen-oxygen double shared covalent bond.
Is O2 soluble in water?
Oxygen is relatively insoluble in water, its solubility being only 264 µM at 25 °C.
An oxygen molecule (O2) is a good example of a molecule with a covalent bond. Ionic bonds occur when electrons are donated from one atom to another. Table salt (NaCl) is a common example of a compound with an ionic bond. You may also learn about a third type of bond.
Is O2 a compound?
The oxygen molecule O2 is considered a molecule but not a compound. This is because O2 is made of two atoms
Why is oxygen considered a polar molecule?
In both molecules, the oxygen atoms attract electrons more strongly than the carbon or hydrogen atoms do, so both molecules have polar bonds.
A: The two oxygen atoms share two pairs of electrons, so two covalent bonds hold the oxygen molecule together.



