Is O2 a compound?
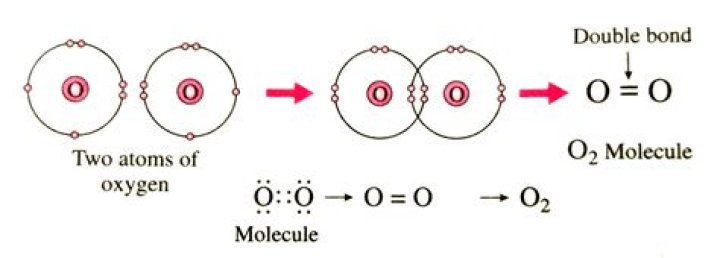
The oxygen molecule O2 is considered a molecule but not a compound. This is because O2 is made of two atoms…
Is O2 still an element?
Oxygen is a chemical element – a substance that contains only one type of atom. Its official chemical symbol is O, and its atomic number is 8, which means that an oxygen atom has eight protons in its nucleus. Two oxygen atoms strongly bind together with a covalent double bond to form dioxygen or O2.
What is O2 compound?
At standard temperature and pressure, two atoms of the element bind to form dioxygen, a colorless and odorless diatomic gas with the formula O2. Diatomic oxygen gas currently constitutes 20.95% of the Earth’s atmosphere, though this has changed considerably over long periods of time.
Molecules have molecular bonds. Oxygen in the atmosphere is a molecule because it contains molecular bonds. It is not a compound because it is made from atoms of only one element – oxygen. This type of molecule is called a diatomic molecule, a molecule made from two atoms of the same type.
Is O2 ionic or covalent?
An oxygen molecule (O2) is a good example of a molecule with a covalent bond. Ionic bonds occur when electrons are donated from one atom to another. Table salt (NaCl) is a common example of a compound with an ionic bond. You may also learn about a third type of bond.
Oxygen is considered an element because it cannot be broken down any farther. Elements are pure substances that form a single atom. Elements are the simplest building blocks you can break matter down into using purely chemical methods. Oxygen can be found on the periodic table with the atomic number eight.
What type of bond is O2?
O2 is a covalent molecule because each oxygen atom needs two valence electrons to complete its octet. To meet this need, each oxygen atom shares two of its electrons with the other oxygen forming a strong oxygen-oxygen double shared covalent bond.
Is CO2 an element?
Carbon dioxide (CO2 ) is a chemical compound composed two oxygen atoms covalentl bonded to a single carbon atom.
No, CO2 is not an ionic compound. Meanwhile, CO2 is a compound that is formed between two non-metal atoms (carbon and oxygen) thus giving it a covalent nature. In CO2 one carbon atom will share its four electrons with two electrons from each of the oxygen atoms.
Is CO2 a covalent compound?
Carbon dioxide is made up of one carbon atom, two oxygen atoms. There are four covalent bonds in one molecule of carbon dioxide. Carbon and oxygen are non-metals, thus we know carbon dioxide is a covalent compound.
Which is not a compound?
compounds are made up of 2 or more DIFFERENT elements. Molecular hydrogen (H2), molecular oxygen (O2) and molecular nitrogen (N2) are not compounds because each is composed of a single element. Water (H2O), carbon dioxide (CO2) and methane (CH4) are compounds because each is made from more than one element. .



