Is NH4 2S a strong electrolyte?
(NH4)2S is a strong electrolyte.
Is NH4 2S a strong electrolyte?
For example, (NH4)2S is soluble and a strong electrolyte. Strong Electrolytes: Essentially all ionic compounds and the strong acids.
Is H2SO4 a strong electrolyte?
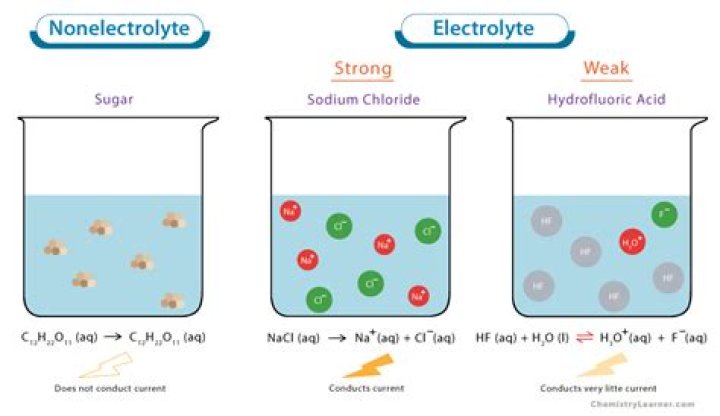
Strong acids and strong bases are strong electrolytes [e.g., HCl(aq), H2SO4 (aq), HClO4(aq); NaOH(aq)]. There are virtually no molecules of a strong acid or base in solution, only ions.
Is NH4OH a weak electrolyte?
answer:NH4OH is a weak electrolyte with dissociation constant equaled to 1.76 10-5. It dissociates partially into ions in an aqueous solution.
Is CH3COOH a weak electrolyte?
Acetic acid is an example of a weak electrolyte even though it is highly soluble in water. A weak electrolyte is an electrolyte that does not completely dissociate in aqueous solution. The solution will contain both ions and molecules of the electrolyte.
Is HNO2 a strong weak or Nonelectrolyte?
Nitrous acid, HNO2(aq), is a weak electrolyte.
Is NH4 strong or weak?
NH4 is a very weak acid since it has an acid dissociation constant of 5.65 * 10. NH4 is a very weak acid since it has an acid dissociation constant of 5.65 * 10.
Is ammonia a weak base?
Ammonia is a typical weak base. Ammonia itself obviously doesn’t contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions. Only about 1% has actually produced hydroxide ions. A weak base is one which doesn’t convert fully into hydroxide ions in solution.
Is CuCl a strong electrolyte?
Salts: Most salts are strong electrolytes. Complex ions, for example Ag(NH3)2+ and CuCl42-, are weak electrolytes.
Is HClO3 a strong electrolyte?
Strong acids are also strong electrolytes. Compounds that are formed from elements of Group 17, such as HCl, HBr and HI, are strong acids. Other strong acids include H2SO4, HNO3, HClO3 and HClO4. Two compounds that are strong electrolytes are the ionic compounds ZnSO4 and CuSO4.
Is nabr a strong electrolyte?
The compound sodium bromide is a strong electrolyte.
What are strong and weak electrolytes give examples?
Thus, this makes acetic acid a weak electrolyte. Carbonic acid (CH2O3), Ammonia (NH3), and Phosphoric acid (H3PO4) act as weak electrolytes. Strong electrolytes examples: Hydrochloric acid (HCl), Sulfuric acid (H2SO4), sodium hydroxide (NaOH), and potassium hydroxide (KOH).
Is nano3 ionic or molecular?
Sodium nitrate features an ionic bond between one Na+ ion and one NO3鈥 ion. The structure of a NaNO3 molecule is illustrated below. The nitrate anion has a trigonal planar structure in which 3 oxygen atoms are bonded to a central nitrogen atom. The negative charge on this ion is delocalized due to resonance.
Is nano3 aqueous or solid?
Sodium nitrate is a white deliquescent solid very soluble in water.



